THERYA NOTES 2026, Vol. 7: 108-114
New Plant Dietary Records for
Two Sympatric Primate Species in Costa Rica
Nuevos Registros de Plantas en la Dieta de
Dos Especies Simpátricas de Primates en Costa Rica
José Manuel Mora1*, Lucía I. López2,3, Leydin García González1, Stephany Cecilia Arce Camacho1, and Dinorah Gámez Chinchilla1
1Carrera de Gestión Ecoturística, Sede Central, Universidad Técnica Nacional (UTN), C. P. 1902-4050. Alajuela, Costa Rica. E-mail: josemora07@gmail.com (JMM), leydin99garci@gmail.com (LGG), arcecamachostephany@gmail.com (SCAC), dinoragamez08@gmail.com (DGC)
2Recinto de Grecia, Universidad de Costa Rica, Tacares, Alajuela, Costa Rica.
3Unidad de Ciencias Básicas y Carrera de Tecnología de Alimentos, Sede Atenas, Universidad Técnica Nacional. Km 34, Ruta 27, C. P. 7-4013. Balsa de Atenas, Costa Rica. E-mail: llopez@utn.ac.cr (LIL).
*Corresponding author
Costa Rica hosts four primate species, including the frugivorous spider monkey Ateles geoffroyi and the omnivorous capuchin Cebus imitator. Although both species are relatively well studied, their diets continue to yield new records. Here we document two previously unreported food plants, a Ficus species consumed by A. geoffroyi and a euphorb eaten by C. imitator, observed during fieldwork in Northwestern Costa Rica. We conducted incidental wildlife observations while hiking the Las Pailas Trail in Rincón de la Vieja National Park, recording all visually or aurally detected vertebrates. For each primate sighting, we noted species, behavior, and foraging context. These records were obtained during academic field visits aimed at documenting natural-history data across contrasting habitats, including dry forest, transitional forest, and geothermal areas. A female spider monkey was observed feeding intensively on Ficus popenoei, consuming at least 20 fruits from a heavily laden tree situated beside an active fumarole. Later, a troop of capuchins fed in a Hura crepitans tree, with one individual dropping a partially eaten fruit. These constitute novel dietary records for A. geoffroyi and C. imitator in Costa Rica. These observations expand the known diets of two Costa Rican primates and highlight their behavioral flexibility. Ateles geoffroyi exploited abundant figs of F. popenoei, a species not previously reported in its diet. Cebus imitator consumed H. crepitans, despite its mechanical and chemical defenses. Such records emphasize the value of natural-history observations for understanding primate ecology and informing conservation in changing landscapes.
Key words: Ateles geoffroyi; Cebus imitator; Ficus popenoei; food habits; Hura crepitans
Costa Rica alberga cuatro especies de primates, entre ellas el frugívoro Ateles geoffroyi y el omnívoro Cebus imitator. Aunque ambas especies están relativamente bien estudiadas, sus dietas continúan aportando nuevos registros. Aquí documentamos dos plantas no reportadas previamente: un Ficus consumida por A. geoffroyi y una euforbiácea ingerida por C. imitator, observadas en el noroeste de Costa Rica. Realizamos observaciones incidentales de fauna mientras recorríamos el sendero Las Pailas en el Parque Nacional Rincón de la Vieja, registrando todos los vertebrados detectados visual o auditivamente. Para cada avistamiento de primates anotamos especie, comportamiento y contexto de forrajeo. Estos registros se obtuvieron durante visitas académicas orientadas a documentar historia natural en hábitats contrastantes, incluidos bosque seco, bosque transicional y zonas geotérmicas. Observamos a una hembra de A. geoffroyi alimentándose de Ficus popenoei, consumiendo al menos 20 frutos de un árbol muy cargado situado junto a una fumarola activa. Más tarde, un C. imitator alimentándose en un árbol de Hura crepitans, dejó caer un fruto parcialmente comido. Estos registros representan nuevos elementos dietarios para A. geoffroyi y C. imitator en Costa Rica. Estas observaciones amplían el conocimiento sobre la dieta de dos primates costarricenses y evidencian su flexibilidad conductual. Ateles geoffroyi explotó la abundancia de frutos de Ficus popenoei, y Cebus imitator consumió Hura crepitans, pese a sus defensas. Estos registros subrayan la importancia de las observaciones de historia natural para comprender la ecología de primates e informar acciones de conservación en paisajes cambiantes.
Palabras clave: Ateles geoffroyi; Cebus imitator; Ficus popenoei; hábitos alimentarios; Hura crepitans
© 2026 Asociación Mexicana de Mastozoología, www.mastozoologiamexicana.org
There are four species of primates in Costa Rica, belonging to two families (Mora and López 2026). The spider monkey, Ateles geoffroyi Kuhl 1820 (Atelidae), ranges from southern Mexico throughout Central America to Panama, typically from sea level to around 1,800 m (Mora and López 2026). The Panamanian white-faced capuchin, Cebus imitator Thomas 1903 (Cebidae), has a more restricted distribution, ranging from Honduras to western Panama and occurring from sea level to about 1,500 m (Rylands and Mittermeier 2013).
The spider monkey occurs primarily in mature and secondary humid forests and mangroves, although it also persists in dry forests such as those of northwestern Costa Rica (Reid 2009). The capuchin monkey, in contrast, inhabits humid lowland, premontane, and montane forests, and also makes frequent use of dry forests, especially gallery forests, and occasionally forages in mangroves (Rylands and Mittermeier 2013). In Costa Rica, it occupies a broad variety of lowland and mid-elevation forest types and has even been recorded above 2,500 m in the Talamanca region (Mora and López 2026).
The spider monkey is among the largest New World primates, measuring 335–582 mm in head-body length and weighing 5.0–9.0 kg, with females generally smaller than males (Reid and Gómez Zamora 2022). Its elongated limbs and highly prehensile, dexterous tail function as a fifth limb for suspension and climbing (Reid 2009; Reid and Gómez Zamora 2022). Ateles geoffroyi exhibits a classic fission–fusion social system in which subgroup size and composition fluctuate according to ecological conditions (Ramos-Fernández and Morales 2014). Subgroups tend to be larger and more cohesive when fruit is abundant (Aguilar-Melo et al. 2018, 2020; Hartwell et al. 2021).
Spider monkeys maintain large home ranges and exhibit exceptional arboreal mobility, allowing them to exploit patchily distributed fruit resources efficiently (Ramos-Fernández and Morales 2014; Aguilar-Melo et al. 2018, 2020). They are among the most frugivorous Neotropical primates, consuming mostly ripe fruits supplemented seasonally with leaves, flowers, seeds, and occasional animal matter (Rylands and Mittermeier 2013; Ramírez-Torres et al. 2025; Mora and López 2026). Their foraging behavior, together with extensive daily movements, makes them important long-distance seed dispersers for many canopy tree species (Rylands and Mittermeier 2013; González‐Zamora et al. 2009).
The spider monkey shows a strong preference for fruits produced by canopy and emergent trees (Chapman 1987). The genus Ficus is particularly important in their diet (Chapman 1987; González-Zamora et al. 2009). In Costa Rica, spider monkeys have been documented feeding on at least 15 Ficus species, with at least eight species recorded at multiple sites (Chaves et al. 2023).
The capuchin monkey is easily recognized by its distinctive coloration: a black crown and uniformly dark body, limbs, and tail contrasted with white facial sides, neck, throat, chest, and shoulders (Mora and López 2026). The abdomen is dark brown, and the tail, densely furred and partially prehensile, provides support during arboreal movement (Reid 2009). This medium-sized primate measures 343–420 mm in head-body length and weighs 2.6–3.9 kg, with females averaging about 27% smaller than males (Rylands and Mittermeier 2013).
Ecologically, the capuchin is a highly adaptable omnivore. Its diet includes fruits, young leaves, buds, flowers, seeds, crabs, snails, clams, eggs, honey, insects and their larvae, and occasionally small vertebrates (Rylands and Mittermeier 2013). It also exhibits specialized prey-handling and food-processing behaviors, such as rubbing caterpillars to remove toxic hairs or spines, sometimes wrapping them in leaves before processing, and following army ants to capture insects flushed from the leaf litter (Panger et al. 2002). Capuchins may also coordinate in small groups when raiding coati nests, with some animals distracting the adults while others seize the young (Rylands and Mittermeier 2013). In tourist-impacted areas, the species is known to modify its foraging patterns and increase intake of human-derived foods (Schulte et al. 2020). Although highly omnivorous, fruit is often the principal dietary component during fruit-rich seasons, while animal prey and other plant items supplement the diet at other times (Rylands and Mittermeier 2013). Capuchins have been documented feeding on a variety of vertebrates, including amphibians, reptiles, birds, and small mammals (Mora et al. 2024). Through their frugivory, capuchins serve as important seed dispersers across tropical forests (Wehncke et al. 2004; Mora and López 2026).
Here we report a previously undocumented Ficus species in the diet of Ateles geoffroyi in Costa Rica, and a new Euphorbiaceae species consumed by Cebus imitator.
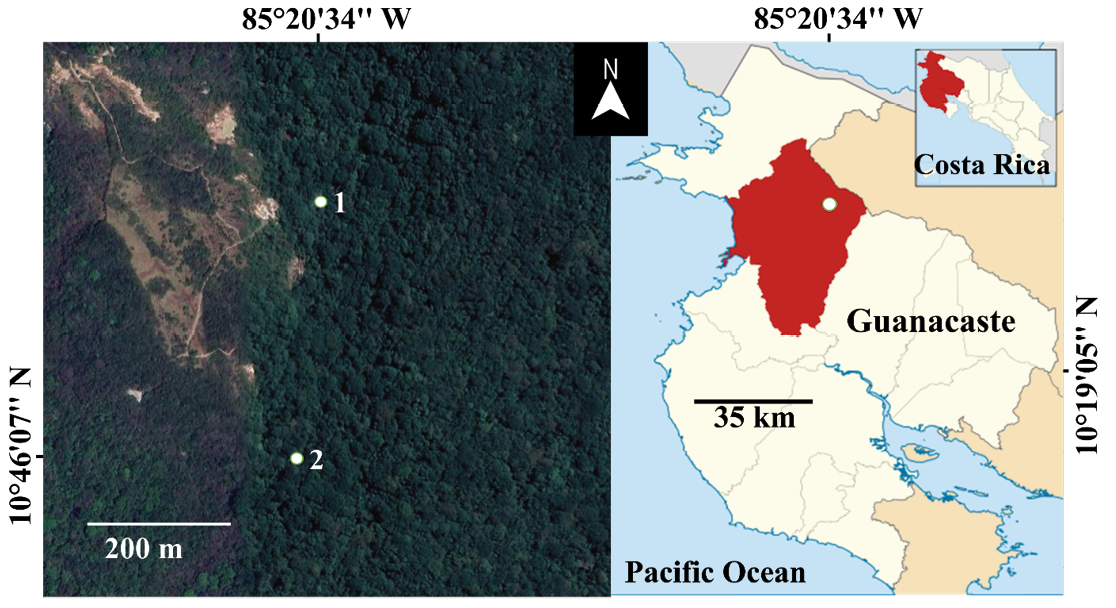
We conducted academic field visits to several protected areas in Costa Rica, hiking sections of the main trails at each site to make natural history observations and to opportunistically document incidental wildlife records, among other objectives. During one of these visits to Rincón de la Vieja National Park (principal crater at 10°49’46” N, 85°19’25” W; Raccichini 2011; Figure 1), located within the Guanacaste Conservation Area, we surveyed the well-established Las Pailas Trail, which traverses dry forest, transitional forest, and areas with active geothermal features.
Along the trail, we recorded all vertebrates detected either visually or aurally, noting species identity, behavior, and habitat context whenever possible. Among the mammals encountered, two of the most notable species were the spider monkey and the Panamanian white-faced capuchin, both frequently observed in the mid- to upper-canopy strata of the park.
The Pailas Trail runs through mature, well-preserved, and protected forest, with short incursions into areas of open vegetation created by volcanic activity, such as sites with fumaroles (Baker 1984; Raccichini 2011). One of our observations was made near one of these sites, while the other occurred within dense forest.
Northwestern Costa Rica, where Rincón de la Vieja National Park is located, is strongly seasonal, with a dry season extending from November/December to April/May (Baker 1984), although it may sometimes last several additional weeks or months (Mora et al. 2015). During the dry season, temperatures reach 30–35 °C in the lower Pacific sections of the park (Baker 1984). Our observations were made toward the end of the rainy season, when deciduous vegetation is still green and many trees retain foliage, while others are producing flowers or fruits (Frankie et al. 1974).
We observed a spider monkey feeding on a fig and a capuchin monkey eating an euphorb fruit. To confirm plant identification, we collected fruit samples directly from the fig tree and retrieved one euphorb fruit dropped by a capuchin monkey.
On 29 November 2025 at 15:19 h, we observed a troop of spider monkeys traveling through the canopy near a fumarole. A solitary female drew particular attention as she fed on the lateral branches of a wild fig tree (Ficus popenoei Standl., Moraceae) growing very close to the geothermal vent (Figure 2). At times, vapors from the fumarole briefly enveloped the tree. We monitored the female for 15 minutes, during which she consumed all or parts of at least 20 figs. We estimated that the tree bore several hundred fruits, with tens visible on a single branch (Figure 3). This was the only tree of this species we observed along the Pailas Trail.
Ficus popenoei is found in moist and wet forests from sea level up to 850 m in several regions of Costa Rica, including the Pacific versant of the Guanacaste Mountain Range (González 2007). As with all members of the genus, F. popenoei engages in an obligate mutualism with its species-specific pollinating fig wasps (Agaonidae or related chalcids), which enter the syconium to pollinate the enclosed inflorescences (Valerio 2004; Machado et al. 2005). In Neotropical forests, the seeds of Ficus species are typically dispersed by frugivorous birds and bats (Valerio 2004).
One distinctive feature of F. popenoei is the soft, velvety pubescence on the undersides of the leaves and on the stipules, giving them a fuzzy texture (González 2007). The leaves are generally large, oval to oblong, and have a thick, coriaceous texture (González 2007). The species produces small, paired syconia (0.9–1.6 × 1.3–2.4 cm) that are also covered in fine hairs (González 2007). These fruits are longer than they are wide and are densely pubescent, with trichomes that range from yellow to golden brown (González 2007).
Later the same day, at 16:15 h, we encountered a troop of capuchin monkeys feeding in the upper canopy of a jabillo tree (Hura crepitans L., Euphorbiaceae). During our observation, one individual dropped a partially eaten fruit (Figure 4). We observed two large jabillo trees along the Pailas Trail: the one where the monkeys were feeding, and another approximately 50 m earlier on the trail, where we saw monkeys, likely from the same troop, passing through the canopy, although none were observed feeding.
Hura crepitans is a large tropical tree recognized for its spiny trunk, caustic latex, and explosively dehiscent seed capsules (Swaine and Beer 1977; Zamora et al. 2004). It occurs primarily in lowland wet tropical forests, riparian zones, and disturbed moist habitats, and commonly reaches heights of 30–40 m (Jiménez et al. 2002). All parts of the plant contain caustic and/or toxic compounds, particularly in the latex and seeds even used to stupefy fish (Hartshorn 1983: Jiménez et al. 2002).
Hura crepitans is a shade-intolerant tree that regenerates primarily in forest-canopy openings (Hartshorn, 1983). Its fruit is a capsule composed of up to sixteen carpels arranged radially around a central axis (Swaine and Beer 1977). The capsule is bright green and somewhat fleshy until fully grown (Swaine and Beer 1977). Mature fruits measure 5–8 cm in diameter (Quesada et al. 1997), resemble small gourds (Figure 4b, c), and ripen about three months after flowering (Little and Wadsworth 1964; Frankie et al. 1974). In Dry Forest habitats, this species exhibits two fruit-drop periods, one in August and another in November (Frankie et al. 1974; Zamora et al. 2004).
The observations presented here provide new insight into the dietary ecology of two Costa Rican primates: Ateles geoffroyi and Cebus imitator and highlight the behavioral flexibility that characterizes both species. Although the natural history and feeding ecology of these primates are relatively well documented, opportunistic field observations continue to reveal previously unreported aspects of their diet, habitat use, and foraging strategies (Mora et al. 2024).
The spider monkey relies heavily on ripe fruits obtained primarily from trees in the upper forest canopy (Chapman 1987). Within this context, figs (Ficus spp.) are important because their asynchronous fruiting patterns provide relatively stable food resources throughout the year (Valerio 2004). Previous studies have documented at least 15 Ficus species consumed by spider monkeys in Costa Rica (Chaves et al. 2023).
Our observation of a female A. geoffroyi feeding extensively on F. popenoei represents, to our knowledge, the first published record of this species serving as a food source for spider monkeys in Costa Rica. Although F. popenoei occurs throughout Mesoamerica and parts of South America, its ecological interactions remain poorly described. The presence of abundant ripe fruits on a tree located adjacent to an active fumarole suggests that even highly localized microhabitat features do not deter spider monkeys when fruit availability is high. The consumption of at least 20 figs in a short period is consistent with the selective and energetically efficient fruit-feeding strategy documented for Ateles geoffroyi, particularly during feeding bouts on Ficus species (Chapman 1987; González-Zamora et al. 2009).
The consumption of Hura crepitans fruit by capuchins is particularly noteworthy given the plant’s suite of defensive traits (Hartshorn 1983). Hura crepitans possesses spines, caustic latex, and seeds contained within explosively dehiscent capsules (Zamora et al. 2004; Vogel 2005). Its latex can cause irritation and skin rashes and may even lead to blindness, while ingestion of the seeds can result in nausea, vomiting, diarrhea, and potentially death (Jiménez et al. 2002). The ability of C. imitator to process and consume these fruits underscores the species’ adaptability and cognitive capacity to exploit challenging food resources. Previous studies have shown that capuchins are capable of handling mechanically or chemically defended foods, frequently employing learned behaviors or socially transmitted techniques to gain access to them (Barrett et al. 2018; Santos et al. 2019; Mora et al. 2024; Rufo et al. 2024). In addition, plant secondary metabolites may confer beneficial effects, including antioxidant and anthelmintic properties, as well as the formation of complexes between proteins and condensed tannins that protect dietary protein from degradation by the symbiotic microflora of foregut fermenters, thereby enhancing protein utilization (Iason 2005).
These opportunistic observations highlight the dietary breadth of both species and reinforce the importance of continued natural-history documentation, even for well-studied taxa. For spider monkeys, identifying additional Ficus species in the diet enhances our understanding of their role as long-distance seed dispersers and their reliance on diverse fruiting phenologies. For capuchins, reports of Hura crepitans consumption contribute to the growing evidence of their adaptability in both natural and human-modified forests.
Given ongoing habitat fragmentation and land-use change in Costa Rica, documenting such behavioral flexibility is essential. Species capable of incorporating a wide array of food types may be better able to persist in disturbed landscapes. Conversely, the strong dependence of spider monkeys on large-canopy fruiting trees, combined with their need for extensive home ranges, underscores their vulnerability to forest loss.
Overall, these observations contribute valuable data to the natural history of Costa Rican primates and emphasize the continued importance of field-based observations for refining our understanding of species ecology, diet, and conservation requirements.
Acknowledgements
We thank two anonymous reviewers for their constructive feedback and valuable suggestions, which helped improve this manuscript. Quírico Jiménez kindly confirmed the identification of the Ficus species. J. M. Mora acknowledges E. Rivera, Department Head of Ecotourism Management (GEC) at the Central Campus of the Universidad Técnica Nacional (UTN), for her academic support.
Literature cited
Aguilar-Melo, A. R., S. Calmé, S. E. Smith-Aguilar, and G. Ramos-Fernandez. 2018. Fission-fusion dynamics as a temporally and spatially flexible behavioral strategy in spider monkeys. Behavioral Ecology and Sociobiology 72: 150.
Aguilar‐Melo, et al. 2020. Ecological and social determinants of association and proximity patterns in the fission–fusion society of spider monkeys (Ateles geoffroyi). Journal of Primatology 82: e23077.
Baker, R. G. 1984. Notes on the flora of Volcan Rincon de la Vieja, Costa Rica. Brenesia 22: 261–283.
Barrett, B. J., et al. 2018. Habitual stone-tool-aided extractive foraging in white-faced capuchins, Cebus capucinus. Royal Society Open Science 5: 81002.
Chapman, C. A. 1987. Flexibility in diets of three primate species. Primates 28: 161–170.
Chaves, Ó.M., et al. 2023. Data and R-Scripts from: Plant diversity in diet of Costa Rican primates in contrasting habitats: A meta-analysis. Figshare Dataset. Retrieved from https://doi.org/10.6084/m9.figshare.21785588.v1.
Di Fiore, A., A. Link, and D. J. Dew. 2008. Diets of wild spider monkeys. Pp. 81–137 in Spider monkeys: Behavior, ecology and evolution of the genus Ateles (Campbell, C. J., ed.). Cambridge University Press, Cambridge, U.K.
Frankie, G. W., H. G. Baker, and P. A. Opler. 1974. Comparative phenological studies of trees in tropical wet and dry forests in the lowlands of Costa Rica. The Journal of Ecology 62: 881–919.
González, J. 2007. Moraceae. Pp. 635–675 in Manual de plantas de Costa Rica. Vol. VI. Dicotiledóneas (Haloragaceae – Phytolaccaceae) (Hammel, B. E., M. H. Grayum, C. Herrera., and N. Zamora, eds). Missouri Botanical Garden Press, St. Louis, Missouri, U.S.A.
González‐Zamora, A., et al. 2009. Diet of spider monkeys (Ateles geoffroyi) in Mesoamerica: current knowledge and future directions. American Journal of Primatology 71: 8–20.
Hartshorn, G. S. 1983. Hura crepitans (Jabillo, Sandbox Tree). Pp. 251–252 in Costa Rican natural history (Janzen, D. H., ed.). University of Chicago Press. Chicago, Illinois, U.S.A.
Hartwell, K. S., et al. 2021. Fruit availability has a complex relationship with fission–fusion dynamics in spider monkeys. Primates 62: 165–175.
Iason, G. 2005. The role of plant secondary metabolites in mammalian herbivory: ecological perspectives. Proceedings of the Nutrition Society 64: 123–131.
Jiménez, Q., F. Rojas, V. Rojas, and L. Rodríguez. 2002. Árboles maderables de Costa Rica Ecología y silvicultura / Timber tres of Costa Rica Ecology and silviculture. INBio. Heredia, Costa Rica.
Little, E. L, Jr., and F. H. Wadsworth. 1964. Common trees of Puerto Rico and the Virgin Islands. U.S Department of Agriculture. Washington, D.C., U.S.A.
Machado, C. A., N. Robbins, and M. T. P. Gilbert. 2005. Critical review of host specificity and its coevolutionary implications in the fig–fig wasp mutualism. Proceedings of the National Academy of Sciences 102: 2435–2440.
Mora, J. M., and L. I. López. 2026. Mamíferos de Costa Rica. Editorial Tecnológica. Cartago, Costa Rica.
Mora, J., F. Rodrigues, L. López, and L. D. Alfaro. 2015. Ctenosaura similis. Cannibalism. Mesoamerican Herpetology 2: 107–109.
Mora, J. M., J. F. Pérez Carrillo, L. Carrillo, and L. I. López. 2024. Comparative behavior of the white-faced capuchin (Cebus imitator) in response to deceased conspecific and non-conspecific infants. Caribbean Journal of Science 54: 121–131.
Panger, M. A., et al. 2002. Cross-site differences in foraging behavior of white-faced capuchins (Cebus capucinus). American Journal of Physical Anthropology 119: 52–66.
Quesada, F. J., Q. Jiménez, N. Zamora, R. Aguilar, and J. González. 1997. Árboles de la Península de Osa. INBio. Heredia, Costa Rica.
Raccichini, S. 1976. Notas sobre la actividad exhalativa cerca del Rincon de la vieja. Costa Rica. Revista Geográfica de América Central 1: 93–104.
Ramírez-Torres, C. E., et al. 2025. Fruit and leaf selection based on plant species and maturity in black-handed spider monkeys (Ateles geoffroyi). Primates 1–11.
Ramos-Fernández, G., and J. M. Morales. 2014. Unraveling fission-fusion dynamics: how subgroup properties and dyadic interactions influence individual decisions. Behavioral Ecology and Sociobiology 68: 1225–1235.
Reid, F. A. 2009. A field guide to the mammals of Central America and Southeast Mexico (2nd ed.). Oxford University Press, Oxford, U.K.
Reid, F., and G. Gómez Zamora. 2022. Pocket guide to the mammals of Costa Rica. Comstock Publishing Associates, Ithaca, U.S.A.
Rufo, H. P., L. G. Ferreira, E. B. Ottoni, and T. Falótico. 2024. Toxic tasting: how capuchin monkeys avoid grasshoppers’ chemical defenses. Primates 65: 235–241.
Rylands, A. B., and R. A. Mittermeier. 2013. Panamanian White-faced Capuchin Cebus imitator. Pp. 412–413 in Handbook of mammals of the world: Vol. 3 Primates (Wilson, D. E., and R. A. Mittermeier, eds.). Lynx Edicions, Barcelona, Spain.
Santos, E. R., et al. 2019. Anointing with commercial insect repellent by free-ranging Cebus capucinus in Manuel Antonio National Park, Quepos, Costa Rica. Primates 60: 559–563.
Schulte, M., G. Gutiérrez-Espeleta, and J. W. Lynch. 2020. Human-monkey interaction dynamics and their dietary impacts on Central American white-faced capuchins (Cebus imitator) at Manuel Antonio National Park, Costa Rica. Neotropical Primates 26: 1–9.
Swaine, M. D., and T. Beer. 1977. Explosive seed dispersal in Hura crepitans L. (Euphorbiaceae). New Phytologist 78: 695–708.
Valerio, C. E. 2004. Los increíbles higuerones / Incredible fig tres. INBio. Heredia, Costa Rica.
Vogel, S. 2005. Living in a physical world II. The bio-ballistics of small projectiles. Journal of Biosciences 30: 167–175.
Wehncke, E. V., C. N. Valdez, and C. A. Domínguez. 2004. Seed dispersal and defecation patterns of Cebus capucinus and Alouatta palliata: consequences for seed dispersal effectiveness. Journal of Tropical Ecology 20: 535–543.
Zamora, N., Q. Jiménez, and J. L. Poveda. 2004. Árboles de Costa Rica / Trees of Costa Rica. Vol. III. INBio. Heredia, Costa Rica.
Associate editor: Romeo A. Saldaña Vázquez
Submitted: December 20, 2025; Reviewed: March 25, 2026
Accepted: April 14, 2026; Published on line: May 8, 2026
DOI: 10.12933/therya_notes-25-239
ISSN 2954-3614
Figure 1. Observation sites (white dots) where two primate species were recorded feeding on fruits in Rincón de la Vieja National Park: (1) the spider monkey Ateles geoffroyi and (2) the white-faced capuchin Cebus imitator. The park is located in Liberia County (highlighted in red), Guanacaste Province (outlined in white and shown in red on the map of Costa Rica). Figure created by José Manuel Mora using imagery from Google Earth (left) and Wikipedia under the Creative Commons Attribution-ShareAlike 3.0 license (right).


Figure 2. Female spider monkey, Ateles geoffroyi, feeding on Ficus popenoei figs in Rincón de la Vieja National Park, Liberia, Guanacaste, Costa Rica. Photo by José Manuel Mora.
Figure 3. Section of the Ficus popenoei tree where a spider monkey, Ateles geoffroyi, was observed feeding on figs in Rincón de la Vieja National Park, Liberia, Guanacaste, Costa Rica. Photo by José Manuel Mora.


Figure 4. a) Capuchin monkey, Cebus capucinus, on a branch of a jabillo tree, Hura crepitans. Photo by José Manuel Mora. b) and c) Two views of a jabillo fruit dropped by the capuchin. Rincón de la Vieja National Park, Liberia, Guanacaste, Costa Rica. Photos by Lucía I. López.