THERYA NOTES 2026, Vol. 7:81-88
Consumption of Myrtillocactus geometrizans
by Frugivorous Bats of the genus Sturnira in a Tropical
Deciduous Forest in the Bajío, Guanajuato, Mexico
Consumo de Myrtillocactus geometrizans por murciélagos frugívoros del género Sturnira en un Bosque
Tropical Caducifolio en el Bajío, Guanajuato, México
Oscar Francisco González-Torres1, Marilyn Vásquez-Cruz1, and Antonio García-Méndez1*
1Tecnológico Nacional de México/ITS de Irapuato. Carretera Irapuato - Silao km. 12, 5 col. El Copal, C.P. 36821, Irapuato, Guanajuato, México. E-mail: oscargleztorres77@gmail.com (OFGT); antonio.gm@irapuato.tecnm.mx (AGM); marilyn.vc@irapuato.tecnm.mx (MVC).
*Corresponding author
Frugivory interactions are essential in tropical ecosystems due to their impact on forest regeneration through seed dispersal. Frugivorous bats play a key role in these interactions because of their ability to travel long distances and consume a wide variety of fruits. This study assessed the diet of frugivorous bats in a tropical deciduous forest (TDF). Bats were captured over an annual cycle from May 2023 to March 2024, covering both the rainy and dry seasons. Four mist nets were set among the vegetation and in natural roosts from 18:00 to 01:00 hours. Captured bats were placed in cloth bags to obtain fecal samples. With a sampling effort of 5760 m/net/night, 35 bats from the families Phyllostomidae, Vespertilionidae, and Molossidae were recorded. Sturnira hondurensis was the most abundant species. Two genera of plants, Solanum and Myrtillocactus, were identified from fecal samples of captured individuals. The presence of garambullo (Myrtillocactus geometrizans) in the diet of S. hondurensis and S. parvidens is reported for the first time. The results suggest that the diet of these bats is composed mainly of Solanum ferrugineum fruits, supplemented with M. geometrizans, reflecting a sequential use of food resources according to plant phenology.
Key words: Bats; El Bajío; frugivory; garambullo; seed dispersal.
Las interacciones de frugivoría son fundamentales en los ecosistemas tropicales debido a su impacto en la regeneración de los bosques mediante la dispersión de semillas. Los murciélagos frugívoros desempeñan un papel clave en estas interacciones al desplazarse grandes distancias y consumir una variedad significativa de frutos. Este estudio evaluó la dieta de murciélagos frugívoros en un Bosque Tropical Caducifolio (BTC). La captura de murciélagos se realizó durante un ciclo anual a partir de mayo de 2023 a marzo de 2024, abarcando la temporada de lluvias y secas. Se instalaron 4 redes de niebla entre la vegetación y en refugios naturales, a partir de las 18:00 a 01:00 horas. Los murciélagos capturados fueron colocados en bolsas de manta para obtener muestras de excretas. Con un esfuerzo de 5,760 m/red/noche, se registraron 35 murciélagos de las familias Phyllostomidae, Vespertilionidae y Molossidae. Sturnira hondurensis fue la especie más abundante. A partir de excretas de los individuos capturados, se identificaron 2 géneros de plantas Solanum y Myrtillocactus. Se reporta por primera vez la presencia del garambullo (Myrtillocactus geometrizans) en la dieta de S. hondurensis y S. parvidens. Los resultados sugieren que la dieta de estos murciélagos se compone principalmente de frutos de Solanum ferrugineum, complementada por M. geometrizans, reflejando un uso temporal de los recursos alimenticios en función con la fenología de las plantas.
Palabras clave: Dispersión de semillas; El Bajío; frugivoría; Garambullo; murciélagos.
© 2026 Asociación Mexicana de Mastozoología, www.mastozoologiamexicana.org
Bats have established important mutualistic relationships with various Neotropical plants, playing a key role in their reproductive biology (Fleming and Heithaus 1981). The fruits consumed by these animals have specific morphological adaptations to attract them, including fruits exposed outside the foliage, inconspicuous colors such as green or brown, and intense or fermented fragrances (Kalko et al. 1996; Fleming 2005).
In tropical and subtropical ecosystems, fruit bats of the family Phyllostomidae are efficient dispersers of pioneer plants (Muscarella and Fleming 2007; Kunz et al. 2011). Their diet includes fruits from the families Moraceae, Piperaceae, Urticaceae, and Solanaceae, with broad interactions across the genera Cecropia, Ficus, Piper, Solanum, and Vismia (Fleming 2004). However, their diet can be influenced by intrinsic and extrinsic factors, including fruit size and availability, inter-specific competition, and ecological habitat factors (Herrera 1989; Karina et al. 2016; Flórez-Montero et al. 2022).
Although this type of interaction has been widely documented in humid tropical areas, it is rare and of little relevance in arid and semi-arid environments, since most studies have focused on humid ecosystems (Heithaus et al. 1975; Heithaus 1982; Galindo-González 1998; Estrada and Coates-Estrada 2001). Numerous studies have addressed the interactions between bats and cacti, although most have focused mainly on nectarivorous species, mainly associated with the pollination of agaves and columnar cacti (Valiente-Banuet et al. 1996; Fleming and Valiente-Banuet 2002; Stoner et al. 2003), although the consumption of their fruits by bats of the genera Sturnira and Artibeus has also been reported (Cornejo-Latorre et al. 2011).
In this context, cacti acquire ecological relevance as a key resource in arid and semi-arid ecosystems. The family Cactaceae comprises approximately 130 genera and about 1850 species that are naturally distributed in the Americas (Nyffeler y Eggli 2010; Hernández-Ledesma et al. 2015; Schwertner-Charão et al. 2022). Mexico stands out as the country with the highest richness of cactus species worldwide, followed by Brazil, Argentina, and Bolivia. Mexico has recorded 670 cactus species, of which 77 % are endemic to the country (Ortega-Baes et al. 2010; Arias and Aquino 2019; Guerrero et al. 2019).
This study reports the first documented consumption of Myrtillocactus geometrizans (Mart. Ex Pfeiff.) Console, 1897 fruits by bats of the genus Sturnira in a tropical deciduous forest in the Bajío region, Guanajuato, Mexico. The garambullo (M. geometrizans) is a species of columnar cactus endemic to Mexico. It has tubular-shaped flowers, with a characteristic greenish-white color, and produces edible berry-like fruits with a blue-reddish hue. Fruiting occurs between March and September, and a single adult plant has been reported to produce around 7650 fruits (Pérez-Villafaña and Valiente-Banuet 2009). The seeds of this species are dispersed mainly by birds and mammals (Bravo-Hollis 1978; Arias 2010).
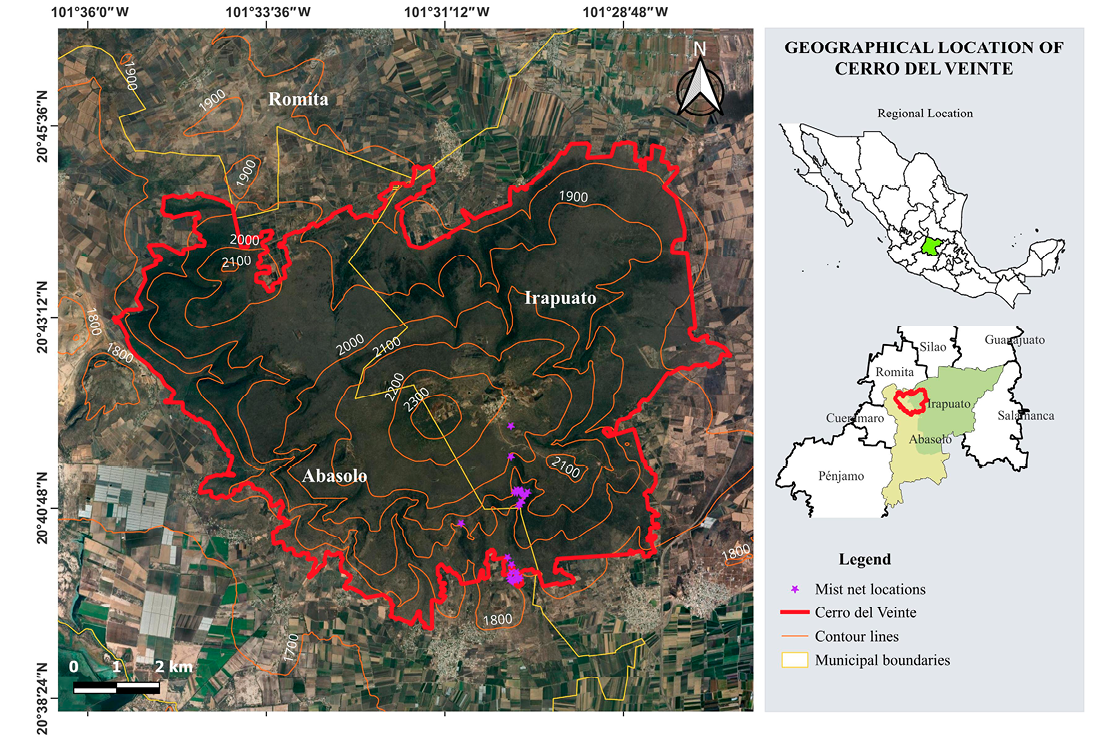
The study was carried out in Cerro del Veinte (20°40’46.61” N, 101°29’50.80” W), located approximately 27 km west of the municipality of Irapuato, bordering the municipalities of Cuerámaro and Abasolo, Guanajuato, within the Bajío region (Figure 1). It covers an approximate area of 6120 ha, with elevations between 1770 m and 2340 m a.s.l. The characteristic vegetation is tropical deciduous forest, with trees 4 m to 10 m high and marked seasonal phenology, along with cacti such as Opuntia and Myrtillocactus. The herbaceous stratum is scarce and grows mainly during the rainy season (Rzedowski and Calderón de Rzedowski 1987; Rzedowski et al. 1996).
The samplings were conducted monthly between May 2023 and March 2024, on two consecutive nights each month, from 6:00 p.m. to 1:00 a.m. In each sampling session, four mist nets (12 m × 3 m) were placed at ground level and at heights up to 3 m, separated by 15 to 20 m. These were set in strategic sites, including areas near crops and zones with the presence of Solanum ferrugineum and M. geometrizans, as well as on dry riverbeds that function as flight corridors. In addition, two caves used as shelters were sampled. The nets were checked every 30 minutes; in the absence of captures, the revision interval was extended by 10 minutes.
The captured bats were handled following the guidelines for the use of wild mammals in research and education established by the American Mammalian Society (Sikes et al. 2016), the biosecurity protocol described in the manual entitled “Investigating the role of bats in emerging zoonoses” issued by the Food and Agriculture Organization of the United Nations (FAO 2011), and “The Field Manual of Bat Handling” published by the Latin American and Caribbean Network for the Conservation of Bats (RELCOM 2020). The captured bats were kept in cloth bags for processing, recording the time of capture, weight (g), sex, reproductive condition, age, and morphometric measurements (forearm) using a digital vernier (Steren HER – 411, China). Species identification was conducted using the field guide by Medellín et al. (2008).
The consumed seeds were collected using two methods: 1) placement of nylon plastic sheets (12 m × 3 m) under the nets (Galindo-González et al. 2009), and 2) temporary retention of bats in cloth bags (20–30 minutes) until defecation (Castro-Luna and Galindo-González 2012). Fecal samples were stored in labeled Eppendorf tubes. In the laboratory, the excreta were rinsed in a Petri dish with chlorinated water solution to remove traces of fruit and pulp. Seeds were counted, sorted by morphotype, dried on absorbent paper, and examined under a trinocular stereo microscope with zoom (Velab VE – S5, Mexico) for identification. The identification was carried out by consulting online herbaria, such as the National Herbarium of Mexico (MEXU), Luz María Villareal de Puga Herbarium (IBUG), Herbarium of the BUAP Botanical Garden (HUAP), and the electronic repository of the Network of Herbaria of Mexico, as well as by comparison with excreted seeds and fruit seeds obtained by direct collection in the field.
Likewise, the germination rate of the seeds excreted by bats was evaluated and compared to the rate of seeds collected directly from fruits. In total, 368 seeds of S. ferrugineum and M. geometrizans were sown. To standardize the experimental design and ensure comparability between excreted and non-excreted seeds, the total number of seeds recovered from feces was quantified in each sample, and the same number of non-excreted seeds obtained directly from fruits were sown. In this way, each excreta sample had its respective reference sample with the same initial number of seeds.
Excreted and non-excreted seeds were placed in Petri dishes with agar-agar culture medium (30 seeds per Petri dish). In cases where the number of seeds exceeded this limit, as many dishes as necessary were used to cover the entire sample. The dishes were maintained and checked daily until radicle growth was observed. The germination percentage was estimated by dividing the number of germinated seeds by the total number of seeds shown per sample.
When a sample failed to germinate during the initial observation period, the seeds were transferred to a new agar medium to extend monitoring and confirm the absence of germination. Subsequently, all the seedlings obtained were transplanted into germination trays and watered daily.
With a sampling effort of 5780 m/net/night, a total of 35 individuals were recorded, belonging to three families and five species, with Phyllostomidae being the most representative family (91.4 %). In contrast, the families Molossidae and Vespertilionidae were represented by only one species each. Catches occurred predominantly in November and December, mainly comprising the genera Choeronycteris and Sturnira (60 % and 31 % of the total catches, respectively). C. mexicana and S. hondurensis were the predominant species, with the latter being present in almost all samplings.
Feces collected from 11 fruit bat specimens yielded 368 seeds of two plant species: S. ferrugineum (Solanaceae) and M. geometrizans (Cactaceae). This study is the first to report the consumption and dispersal of M. geometrizans seeds (Figure 2 a, b, c), constituting a new record of the trophic interaction between fruit bats of the genus Sturnira and this columnar cactus in a tropical deciduous forest in the Bajío region.
Regarding the frequency of occurrence, S. hondurensis presented the highest proportion of feces samples with seeds (83 %), in addition to being the only species in which the simultaneous presence of seeds of S. ferrugineum (97 %) and M. geometrizans (3 %) was recorded. In the case of S. parvidens, although less frequently captured, the presence of M. geometrizans seeds was also observed; these were the only seeds collected for this species. These findings indicate that both species of the genus Sturnira actively participate in the consumption and, probably, in the dispersal of M. geometrizans seeds.
The germination assay showed marked differences between excreted and non-excreted seeds. Of six samples of excreted seeds, two germinated at 10 days (21 % for M. geometrizans and 2 % for S. ferrugineum), and a third from S. ferrugineum germinated at 20 days. The remaining seeds failed to germinate, even after being transferred to a new medium. In comparison, non-excreted seeds achieved 10 % germination at 10 days, compared to 21% of excreted seeds, suggesting a positive influence on the seed dispersal process and, potentially, on vegetation regeneration in the studied area (Figure 3).
This work reports for the first time the presence of M. geometrizans in the diet of two fruit bats of the genus Sturnira in a tropical deciduous forest in the Bajío region, Mexico. This finding represents a new trophic interaction between these bats and a columnar cactus and expands our knowledge about bat-plant interactions in arid and semi-arid regions.
This record coincides with the bat catch peaks observed during the transition between the dry and wet seasons, in which fruit production and availability increase, while the lowest catches occurred in the cold season, possibly due to lower foraging intensity (Sánchez-Cordero 2001; Bejarano-Bonilla et al. 2007; Ribeiro-Mello 2009; Estrada-Villega 2010).
In terms of diet, S. ferrugineum seeds were the most abundant in excreta, consistent with previous studies that identified species of the genus Solanum as a key component in the diet of fruit bats of the genus Sturnira (Heithaus et al. 1975; Medellín and Gaona 1999; Salvador and Yurrita 2005). Solanum fruits exhibit chiropterochory-syndrome characteristics (Poulin and McNeil 1992), which may explain this preference. In contrast, M. geometrizans represents a resource reported for the first time in the Sturnira diet. This interaction suggests high dietary flexibility, in which bats exploit alternative resources in response to seasonal availability, as proposed by Heithaus (1982) and Fleming (1986). The presence of these seeds indicates that Sturnira not only consumes them but potentially contributes to their effective dispersal (Dumont 1999).
Although interactions between bats and cacti have been widely documented, most studies have focused on nectarivorous species of the genera Leptonycteris, Choeronycteris, and Glossophaga, mainly associated with the pollination of agaves and columnar cacti (Valiente-Banuet et al. 1996; Fleming and Valiente-Banuet 2002; Stoner et al. 2003). However, it has been observed that the fruits of some cacti are consumed by fruit bats, as recorded here for the genus Sturnira. In this context, our record represents an important contribution by demonstrating that Sturnira interacts with the fruits of M. geometrizans and has the potential to act as a seed disperser, providing supplementary ecological functions in these ecosystems (Godinez-Álvarez and Valiente-Banuet 2000; Sánchez-Casas and Álvarez 2000; Cornejo-Latorre et al. 2011).
Given that M. geometrizans is a columnar cactus endemic to Mexico with a high fruit production during several months of the year (Pérez-Villafaña and Valiente-Banuet 2009), its use by fruit bats could represent an alternative feeding strategy with mutual benefits for both species. In disturbed or fragmented vegetation, these interactions could favor natural regeneration, particularly in the tropical deciduous forest, where zoochoric dispersal is essential.
In addition, our results show that passage of seeds through the digestive tract of bats increases their germination, consistent with observations by Traveset and Verdú (2002). In M. geometrizans, excreted seeds had higher germination percentages than seeds obtained directly from the fruit, a pattern similar to that described by Naranjo et al. (2003) for Stenocereus and Subpilocereus species. This same pattern was also observed in the study by Pérez-Villafaña and Valiente-Banuet (2009), where the seeds of M. geometrizans ingested by Sturnira lilium reached 75 % germination, a figure considerably higher than that for non-ingested seeds (15 %). These differences may be associated with the experimental conditions or the selection and manipulation of fruits by bats (Dumont 1999). Therefore, we recommend replicating the germination experiment with a larger number of seeds and a more rigorous control of the germination conditions.
Acknowledgements
We thank the Tecnológico Nacional de México/ITS Irapuato for the institutional support provided for the conduct of this work, through the financial support of the project “Characterization and Analysis of the Diet of Medium and Flying Mammals in Natural and Disturbed Areas” granted to the third author. We also express our sincere gratitude to the anonymous reviewers for their valuable comments, which significantly improved this manuscript.
Literature cited
Arias, S. 2010. Cactáceas comestibles. Instituto de Biología, Jardín Botánico de la Universidad Nacional Autónoma de México. Ciudad de México, México. 142 pp.
Arias, S. and D. Aquino. 2019. Flora del Bajío y de regiones adyacentes familia Cactaceae I. Instituto de Ecología, A.C. Pátzcuaro, México. 278 pp.
Bejarano-Bonilla, D. A. et al. 2007. Bat diversity and distribution along an altitudinal transect in the Tolima region of Colombia. Caldasia 29:297–308.
Bravo-Hollis, H. and H. Sánchez - Mejorada. 1978. Las cactáceas de México. Universidad Nacional Autónoma de México. Ciudad de México, México. 643 pp.
Castro-Luna, A. A., and J. Galindo-González. 2012. Seed dispersal by phyllostomid bats in two contrasting vegetation types in a Mesoamerican reserve. Acta Chiropterologica 14:133–142.
Cornejo-Latorre, C. et al. 2011. Abundancia estacional de los murciélagos herbívoros y disponibilidad de los recursos quiropterófilos en dos tipos de vegetación de la Reserva de la Biosfera Barranca de Metztitlán, Hidalgo, México. Therya 2:169–182.
Dumont, E. R. 1999. The effect of food hardness on feeding behaviour in frugivorous bats (Phyllostomidae): an experimental study. Journal of Zoology 248:219–229.
Estrada, A. and R. Coates-Estrada. 2001. Species composition and reproductive phenology of bats in a tropical landscape at Los Tuxtlas, Mexico. Journal of Tropical Ecology 17:627–646.
Estrada-Villega, M. 2010. Ensamblaje de murciélagos en un bosque subandino colombiano y análisis sobre la dieta de algunas especies. Mastozoología Neotropical 17:31–41.
Food and Agriculture Organization of the United Nations (FAO). 2011. Investigating the role of bats in emerging zoonoses: balancing ecology, conservation and public health interests (S. H. Newman, H. Field, J. Epstein and C. de Jong, eds.). FAO Animal Production and Health Manual No. 12. Food and Agriculture Organization of the United Nations. Roma, Italia. 169 pp.
Fleming, T. H. 1986. The structure of neotropical bat communities: a preliminary analysis. Revista Chilena de Historia Natural 59:135–150.
Fleming, T. H. and Heithaus, E. R. 1981. Frugivorous bats, seed shadows, and the structure of tropical forests. Biotropica 13:45–53.
Fleming, T. H. 2004. Dispersal ecology of neotropical Piper shrubs and treelets. Pp. 58-77 in Piper: a model genus for studies of phytochemistry, ecology, and evolution (Dyer, L. A. and A. D. N. Palmer, eds.). Springer. Boston, EE.UU.
Fleming, T. 2005. The relationship between species richness of vertebrate mutualists and their food plants in tropical and subtropical communities differs among hemispheres. Oikos 111:556–562.
Fleming, T. H. and A. Valiente-Banuet. 2002. Columnar cacti and their mutualists: evolution, ecology and conservation. University of Arizona Press, Tucson, EE.UU. 371 pp.
Flórez-Montero, G. L. et al. 2022. NeoBat interactions: a data set of bat–plant interactions in the Neotropics. Ecology 103:e3640.
Galindo-González, J. 1998. Dispersión de semillas por murciélagos: su importancia en la conservación y regeneración del bosque tropical. Acta Zoológica Mexicana 74:57–74.
Galindo-González, J., G. Vázquez-Domínguez, R. A. Saldaña-Vázquez and J. R. Hernández-Montero. 2009. A more efficient technique to collect seeds dispersed by bats. Journal of Tropical Ecology 25:205–209.
Godínez-Álvarez, H. and A. Valiente-Banuet. 2000. Fruit-feeding behavior of the bats Leptonycteris curasoae and Choeronycteris mexicana in flight cage experiments: consequences for dispersal of columnar cactus seeds. Biotropica 32:552–556.
Guerrero, P. C. et al. 2019. Phylogenetic relationships and evolutionary trends in the cactus family. Journal of Heredity 110:4–21.
Heithaus, E. R. 1982. Coevolution between bats and plants. Pp. 243–264 in Ecology of bats (Kunz, T. H., ed.). Springer. Boston, EE.UU.
Heithaus, E. R., T. H. Fleming and P. A. Opler. 1975. Foraging patterns and resource utilization in seven species of bats in a seasonal tropical environment. Ecology 56:841–854.
Hernández-Ledesma, P. et al. 2015. A taxonomic backbone for the global synthesis of species diversity in the angiosperm order Caryophyllales. Willdenowia 45:281–383.
Herrera, C. M. 1989. Frugivory and seed dispersal by carnivorous mammals, and associated fruit characteristics, in undisturbed Mediterranean habitats. Oikos 55:250–262.
Karina, A. et al. 2016. Diet preference in frugivorous bats (Phyllostomidae) within a fragment of dry tropical forest. Revista de Ciencias 20:139–146.
Kalko, E. K. V, Handley, C. O. and Handley, D. 1996. Chapter 16 Organization, Diversity, and Long-Term Dynamics of a Neotropical Bat Community. Pp. 503-553 in Long-Term Studies of Vertebrate Communities (Cody, M. L. and J. A. Smallwood, eds.) Academic Press. San Diego, EE.UU.
Kunz, T., Braun de Torrez, E., Bauer, D., Lobova, T. and Fleming, T. 2011. Ecosystem services provided by bats. Annals of the New York Academy of Sciences 1223:1–38.
Medellín, R. A. and O. Gaona. 1999. Seed dispersal by bats and birds in forest and disturbed habitats of Chiapas, Mexico. Biotropica 31:478–485.
Medellín, R., H. Arita and O. Sánchez. 2008. Identificación de los murciélagos de México. Clave de campo. Segunda edición. Instituto de Ecología, Universidad Nacional Autónoma de México. México, México. 79 pp.
Muscarella, R. and T. H Fleming. 2007. The role of frugivorous bats in tropical forest succession. Biological reviews 82:573-590.
Naranjo, M. E., C. Rengifo and P.J. Soriano. 2003. Effect of ingestion by bats and birds on seed germination of Stenocereus griseus and Subpilocereus repandus (Cactaceae). Journal of Tropical Ecology 19:19–25.
Nyffeler, R. and U. Eggli. 2010. A farewell to dated ideas and concepts: molecular phylogenetics and a revised suprageneric classification of the family Cactaceae. Schumannia 6:109–149.
Ortega - Baes, P. et al. 2010. Diversity and conservation in the cactus family. Pp. 157–173 in Desert plants: biology and biotechnology (Ramawat, K. G., ed.). Springer-Verlag. Berlin, Alemania.
Pérez-Villafaña, M. G. and A. Valiente-Banuet. 2009. Effectiveness of dispersal of an ornithocorous cactus Myrtillocactus geometrizans (Cactaceae) in a patchy environment. The Open Biology Journal 2:101–113.
Poulin, B. and R. McNeil. 1992. Tropical avian phenology in relation to abundance and exploitation of food resources. Ecology 73:2295–2309.
RELCOM (Red Latinoamericana y del Caribe para la Conservación de los Murciélagos). 2020. Manual de manejo de murciélagos en campo. Disponible en: www.relcomlatinoamerica.net/images/PDFs/Manual_de_manejo_de_murcielagos.pdf . Consultado el 1 de diciembre de 2025.
Rzedowski, J. and G. Calderón de Rzedowski. 1987. El bosque tropical caducifolio de la región mixteca del Bajío. Trace 12:12–21.
RzedowskI, J., G. Calderón de Rzedowski and R. Galván. 1996. Nota sobre la vegetación y la flora de la zona noreste del estado de Guanajuato. Flora del Bajío y Regiones Adyacentes 14:1–22.
Ribeiro-Mello, M. A. 2009. Temporal variation in the organization of a Neotropical assemblage of leaf-nosed bats (Chiroptera: Phyllostomidae). Acta Oecologica 35:280–286.
Salvador, L. and C. L. Yurrita. 2005. Análisis de nicho alimentario en la comunidad de murciélagos frugívoros de Yaxhá, Petén, Guatemala. Acta Zoológica Mexicana 21:83–94.
Sánchez-Casas, N. and T. Álvarez. 2000. Palinofagia de los murciélagos del género Glossophaga (Mammalia: Chiroptera) en México. Acta Zoológica Mexicana (n.s.) 81:23–62.
Sánchez-Cordero, V. 2001. Elevation gradients of diversity for rodents and bats in Oaxaca, Mexico. Global Ecology and Biogeography 10:63–76.
Schwertner-Charão, L. et al 2022. Las fascinantes adaptaciones de las cactáceas y su historia evolutiva. CIENCIA Ergo-Sum, 30:1-7.
Sikes, R. S. et al 2016. Guidelines of the American Society of Mammalogists for the use of wild mammals in research and education. Journal of Mammalogy 97:663–688.
Stoner, K. E. et al. 2003. Population dynamics, reproduction, and diet of the lesser long-nosed bat (Leptonycteris curasoae) in Jalisco, Mexico: implications for conservation. Biodiversity and Conservation 12:357–373.
Traveset, A. and Verdú, M. 2002. A meta-analysis of the effect of gut treatment on seed germination. Pp. 339-350 in Seed Dispersal and Frugivory: Ecology, Evolution and Conservation (Levey, D., W. Silva and M. Galetti, eds.). CABI Publishing. Wallingford, Reino Unido.
Valiente-Banuet, A. et al. 1996. Ecological relationships between columnar cacti and nectar-feeding bats in Mexico. Journal of Tropical Ecology 12:103–119.
Associate editor: Beatríz del S. Bolívar Cimé
Submitted: August 08, 2025; Reviewed: January 08, 2026
Accepted: January 20, 2026; Published on line: April 23, 2026
DOI: 10.12933/therya_notes-25-232
ISSN 2954-3614

Figure 1. Location of the study area and bat capture sites in Cerro del Veinte, Irapuato, Guanajuato, Mexico.

Figure 2. Fruit and seed of Myrtillocactus geometrizans: a) Fruit of M. geometrizans, b) seed of M. geometrizans excreted by the fruit bat (Sturnira), and c) seed excreted in bat feces with seedling in an early development stage.
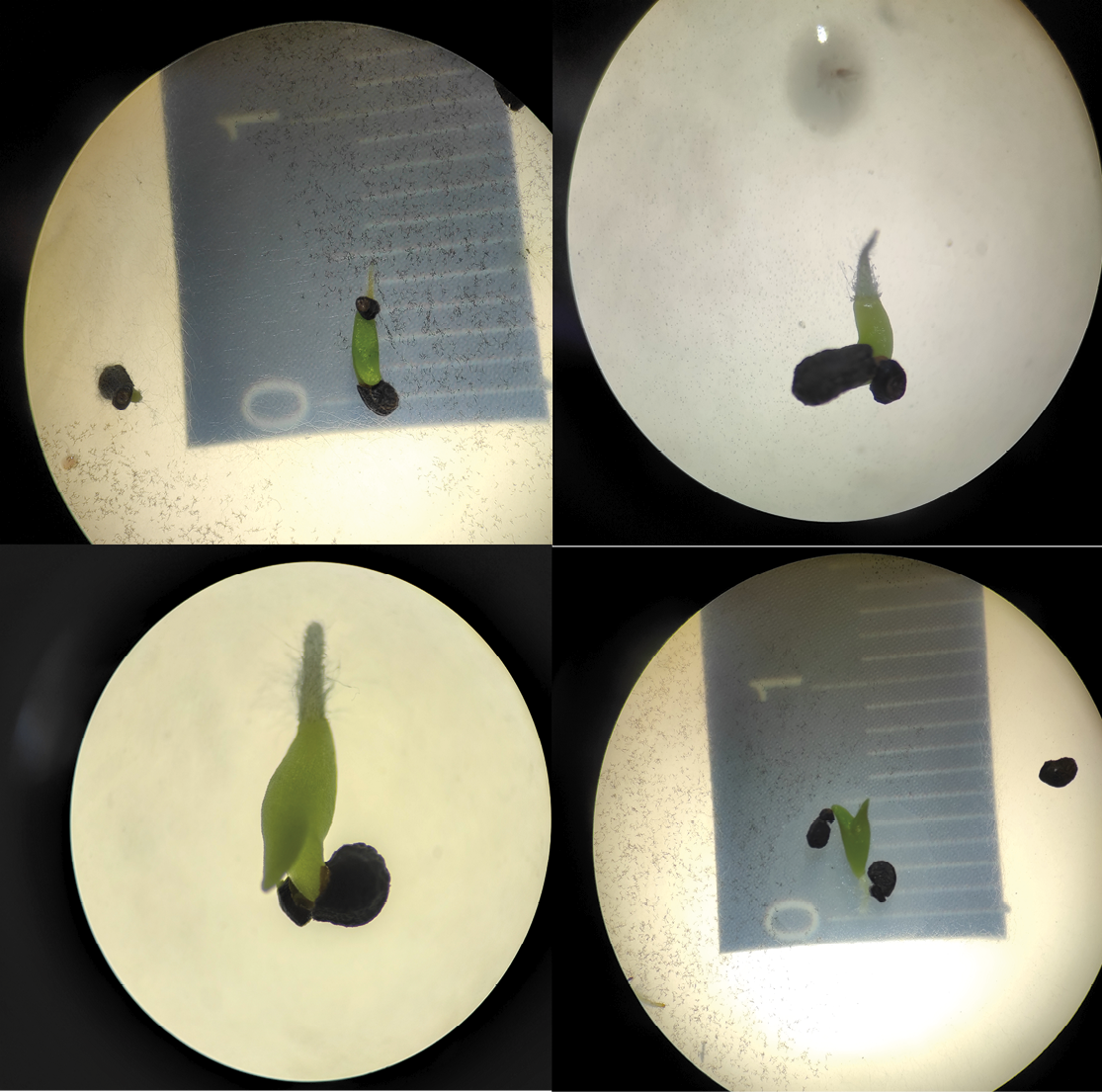
Figure 3. Germination test of Myrtillocactus geometrizans seeds excreted by fruit bats (Sturnira) grown in Petri dishes with agar-agar medium.