THERYA NOTES 2026, Vol. 7:45-50
Morphological description of the lingual surface of Platyrrhinus infuscus under scanning electron microscopy (SEM)
Descripción morfológica de la superficie lingual de Platyrrhinus infuscus mediante microscopía electrónica de barrido (MEB)
Paula Andrea Becerra-Díaz1,3,4,5*, Carlos Bravo-García2,4,5,6, Abelardo Rodríguez-Bolaños1,3,4,5, and Freddy Rodríguez-Saza3,6
1Museo de Historia Natural Universidad Distrital – MHNUD, Universidad Distrital Francisco José de Caldas, Carrera 4A 26 D-54, C.P. 110311, Bogotá, D.C., Colombia. E-mail: pabecerrad@udistrital.edu.co (PB-D); arodriguezb@udistrital.edu.co (AR-B).
2Instituto de Investigación de Recursos Biológicos Alexander von Humboldt, Cra. 8-152, CP. 154001, Villa de Leyva; Boyacá., Colombia. E-mail: caabravog@udistrital.edu.co (CB-G).
3Programa Académico de Biología, Facultad de Ciencias Matemáticas y Naturales, Universidad Distrital Francisco José de Caldas, Carrera 4A 26 D-54, CP. 110311, Bogotá, D.C., Colombia. E-mail: pabecerrad@udistrital.edu.co (PB-D); arodriguezb@udistrital.edu.co (AR-B); frodriguezs@udistrital.edu.co (FR-S).
4Grupo de Investigación de Biodiversidad de Alta Montaña – BAM, Universidad Distrital Francisco José de Caldas, Carrera 4A 26 D-54, CP. 110311, Bogotá, D.C., Colombia. E-mail: pabecerrad@udistrital.edu.co (PB-D); caabravog@udistrital.edu.co (CB-G); arodriguezb@udistrital.edu.co (AR-B).
5SICOM Semillero de Investigación en Conservación y Ecología de Mamíferos, Universidad Distrital Francisco José de Caldas, Carrera 4A 26 D-54, CP. 110311, Bogotá, D.C., Colombia. E-mail: pabecerrad@udistrital.edu.co (PB-D); caabravog@udistrital.edu.co (CB-G); arodriguezb@udistrital.edu.co (AR-B).
6PhyloGenomicsUD – Semillero de Investigación, Creación e Innovación en Filogenética, Evolución y Ciencias ómicas, Grupo de Investigación MATTOPO, Universidad Distrital Francisco José de Caldas, Carrera 4A 26 D-54, CP. 110311, Bogotá, D.C., Colombia. E-mail: caabravog@udistrital.edu.co (CB-G); frodriguezs@udistrital.edu.co (FR-S).
*Corresponding author
La morfología lingual en murciélagos constituye una estructura clave vinculada a sus hábitos alimentarios, aunque en varias especies neotropicales sigue siendo poco explorada. En este trabajo se describe la superficie lingual de Platyrrhinus infuscus mediante el uso de Microscopía Electrónica de Barrido (MEB). Se analizó tejido proveniente de un ejemplar depositado en la colección del Museo de Historia Natural de la Universidad Distrital Francisco José de Caldas (MHNUD). Las papilas fueron clasificadas siguiendo las categorías generales descritas en la literatura: circunvaladas, fungiformes, cónicas y filiformes. Se reconocen once tipos de papilas: circunvaladas; fungiformes con morfología triangular, circular y en cúspide; cónicas; filiformes; gigantes tricúspides y bicúspides; córneas; filiformes estrictas; digitiformes y bífidas. Las papilas de la región de raíz mostraron orientación lateral, mientras que en el ápice se dispusieron hacia la región medial de la lengua. La disposición topográfica de las papilas refleja el trayecto del alimento: las estructuras del ápice facilitan la fijación de frutos carnosos durante el vuelo, mientras que las papilas queratinizadas favorecen la retención y deglución. La zona posterior, dominada por papilas circunvaladas y fungiformes, estaría asociada a una mayor percepción sensorial. Los patrones observados refuerzan la relación de P. infuscus con una dieta frugívora e insectívora, aportando elementos funcionales de interés para investigaciones ecológicas y taxonómicas en murciélagos neotropicales.
Palabras clave: Chiroptera; Microscopía Electrónica de Barrido; morfología lingual; papilas; Phyllostomidae.
Lingual morphology in bats represents a key structure linked to their feeding habits, although it remains poorly explored in several Neotropical species. This study describes the tongue surface of Platyrrhinus infuscus using Scanning Electron Microscopy (SEM). Tissue from a specimen deposited in the collection of the Museo de Historia Natural de la Universidad Distrital Francisco José de Caldas (MHNUD) was analyzed. The papillae were classified according to the general categories described in the literature: circumvallate, fungiform, conical, and filiform. Eleven types of papillae were identified: circumvallate; fungiform with triangular, circular, and cusp-shaped morphologies; conical; filiform; giant tricuspid and bicuspid; horny; strictly filiform; digitiform; and bifid. Papillae in the root region showed lateral orientation, while those at the apex were directed toward the medial region of the tongue. The topographic arrangement of the papillae reflects the food trajectory: structures at the apex facilitate the fixation of fleshy fruits during flight, whereas keratinized papillae promote retention and swallowing. The posterior zone, dominated by circumvallate and fungiform papillae, appears to be associated with enhanced sensory perception. The observed patterns reinforce the association of P. infuscus with a frugivorous and insectivorous diet, providing functional evidence of interest for ecological and taxonomic studies of Neotropical bats.
Key words: Chiroptera; lingual morphology; papillae; Phyllostomidae; Scanning Electron Microscopy.
© 2026 Asociación Mexicana de Mastozoología, www.mastozoologiamexicana.org
DOI: 10.12933/therya_notes-25-230
ISSN 2954-3614
Chiroptera is one of the most diverse mammalian orders in Colombia, with 222 recognized species exhibiting a broad range of trophic niches, including a wide variety of food items such as small mammals, birds, amphibians, reptiles, insects, fish, blood, fruits, pollen, and nectar (Calonge 2009; Cerón-Hernández et al. 2022; Ramírez-Chaves et al. 2024). A fundamental aspect of mammalian evolution is related to feeding mechanisms (Dumont et al. 2012; Iwasaki 2022). For bats, the correlation between functional aspects of the skull, teeth and tongue with the different trophic guilds of the group has been extensively evaluated (Hill and Smith 1984; Fleming and Muchhala 2008; Dumont et al. 2012; González-Terrazas et al. 2012).
Anatomically, the tongue is a muscle that is one of the organs related to feeding and its importance in lingual echolocation has even been evaluated in bats of the genus Rousettus (Roberts 1975; Harper et al. 2013; Attaallah et al. 2023; Lee et al. 2017; Weinberg et al. 2023). In addition, the tongue plays a key role in both sensory and mechanical functions, since mechanical and taste buds constitute part of its surface epithelium. Several studies have demonstrated that the distribution of different types of papillae on the lingual surface varies between species and among individuals of the same species (Abumandour and El-Bakary 2013; Gregorin and Zanatta 2021). On the other hand, it has been suggested that the tongue of bats, being an organ with specialized patterns and microstructures that provide greater efficiency in food consumption, shows a close correlation between its morphology and diet, and even with phylogeny (Sonntag 1926; Doran 1975; Trzcielińska‐Lorych et al. 2009; Abumandour and El-Bakary 2013). However, lingual morphology as a character related to trophic specialization remains an underexplored field.
The few studies conducted in the Neotropics have focused on the following families: Phyllostomidae, Molossidae, Noctilionidae (Gregorin 2003; Masuko et al. 2007), as well as Emballonuridae, Furipteridae, Thyropteridae, Mormoopidae, Natalidae and Vespertilionidae (Gregorin and Zanatta 2021). These investigations have enabled the evaluation of lingual anatomy as a morphological, diagnostic and phylogenetic reference (Birt et al. 1997; Elizalde-Arellano et al. 2004a). However, no such studies have been carried out in Colombia, despite its potential relevance for ecology, taxonomy, and physiology.
Platyrrhinus infuscus is a species of the family Phyllostomidae that is widely distributed in Colombia, Brazil, Bolivia, Ecuador, and Peru, mainly at medium and high elevation (Gardner 2008). Despite its wide distribution, the available information on its feeding ecology remains scarce. It is recognized as a predominantly frugivorous species, with records of consumption from Moraceae (Ficus spp.) and Solanaceae, as well as occasional insectivorous and nectarivorous habits (Rojas et al. 2011; Arias and Pacheco 2019). Nevertheless, current knowledge highlights notable gaps when compared with other widely studied Neotropical frugivores, such as Artibeus lituratus or Carollia perspicillata (Estrada-Villegas et al. 2019), which hinders accurate assessments of its ecological role in forest dynamics.
This study of P. infuscus is significant because it combines dietary and morphological characteristics that differentiate it from other sympatric frugivores. Its association with Ficus spp. fruits, a key resource due to their year-round availability in the tropics (Shanahan et al. 2001), indicates an important role in seed dispersal and, consequently, in the regeneration of degraded forests. Likewise, the specialized tongue morphology, with the presence and arrangement of papillae adapted to fruit processing, represents a functional trait directly linked to its feeding ecology. Considering that tongue morphology in frugivorous bats has been associated with the ability to manipulate and exploit different types of fruit (Winter and von Helversen 2003), this study—being the first conducted in Colombia—provides a detailed description of the tongue surface of P. infuscus through the analysis of images obtained by scanning electron microscopy (SEM). Not only does it fill a gap in the literature at the morphological level, but it also contributes to morphological, taxonomic, ecological, physiological, and phylogenetic studies of P. infuscus.
The sample under study was obtained from a specimen of Platyrrhinus infuscus that is part of the collection of the Museo de Historia Natural de la Universidad Distrital Francisco José de Caldas (MHNUD) (Rodríguez-Bolaños 2020), catalogued under the code MUD-DOC M18. The specimen was previously identified through comparison with other specimens in the collection and by applying the taxonomic keys of Díaz et al. (2021) for Neotropical bats.
The tongue was dissected from the individual by taking as reference the origin of the trachea and performing an incision at the thoracic level over, above the sternum. The cut followed the midline of the abdomen down to the lower left quadrant and then extended to the right, forming a triangle. To ensure complete exposure and visibility of the internal organs, an incision was made at the basal part of the trachea, along with small cuts in the buccopharyngeal muscles that support the tissue. Once the organ was extracted, it was rehydrated through successive washes with distilled water. The sample was stored in a 1.5 ml Eppendorf tube with 90% alcohol for its subsequent analysis, and once the analysis was completed, it was returned to the scientific collection of the MHNUD, as part of the specimen.
In the initial analysis, the general aspects of the morphology of the lingual surface were described using stereoscopic observation (Zeiss) and quantitative data were recorded with a conventional caliper. Microscopic photomicrographs were obtained at the Testing and Assay Laboratory of the Pontificia Universidad Javeriana in Bogotá, the tongue was subjected to critical point drying for 90 minutes and then coated with palladium-gold using a sputter coater (Q150R, QuorumTech). Subsequently, high-resolution images of the papillary morphology of the tongue were captured with a scanning electron microscope (SEM, Zeiss Evo HD15) at 20 kV in high-vacuum mode.
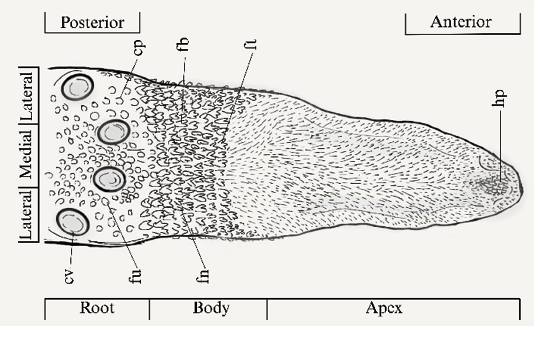
The nomenclature was established based on the distribution of papillae described in the literature, the tongue was divided into three regions for analysis: apex (anterior, medial and lateral apex), body (medial and lateral region) and root (lateral, medial and posterior region) (Gunawan et al. 2020) (Figure 1). For the classification of the papillae, the adjusted nomenclature proposed by Gregorin (2003) was applied.
Macroscopically, the tongue is flat, elongated to a length of 18 mm and a rounded apex. Initially, four types of lingual papillae were identified: circumvallate and fungiform (gustatory), as well as conical and filiform (mechanical). Six subtypes of filiform papillae were found and three subtypes of fungiform papillae. In the posterior region of the root there are four circumvallate papillae (cv): two in the medial region, one in the right lateral region, and one in the left lateral region, all showing grooves and poorly developed epithelia in the surrounding area (Figure 2a).
Next to the circumvallate papillae there are circular fungiform papillae (fu) located in the posterior zone. Additionally, conical papillae (cp) are arranged towards the anterior zone of the root and the middle body, followed by giant bicuspid (fb) and tricuspid filiform papillae (ft), which are distributed across the transitional zone between the root and the body. Tricuspid giant filiform papillae were less numerous than bicuspids (Figure 2a).
In the body region, proximal to the root, numerous bicuspid and giant tricuspid filiform papillae were observed under stereoscopy. These papillae are robust, with central grooves, and arranged in the medial and lateral areas, accompanied by fungiform papillae forming a triangular pattern (Figure 2b). On the lingual borders, some fungiform papillae (mushroom or dome shaped) were also observed; in this region, it was evident that the bicuspid papillae decrease in size (Figure 2c). In the intermediate zone between the body and the apex, strictly filiform papillae (fe) with an elongated form, composed of two or more projections oriented towards the midline in the direction of the pharynx, were distributed (Figure 2d).
At the lingual apex there is evidence of an island of horny papillae (hp), large size and divided medially, surrounded by digitiform papillae (dt) of a smaller size, each consisting of more than five processes. The bifid papillae (fi) appear thick, with a V-shaped or two-pointed apex and only a few projections (Figure 2e). Strictly filiform papillae are distributed across the entire apex, surrounding the other papillae. They are also present along the lingual edges, extending laterally and posteriorly towards the ventral plane. The orientation of this entire set of papillae is directed towards the medial region of the body area (Figure 1).
The morphological characteristics of fruit bats suggest that the arrangement, quantity, location, shape, and microstructures of the lingual papillae contribute to increased feeding efficiency (Birt et al. 1997). The first type of papillae that come into contact with food are the filiform papillae, with at least 16 recognized subtypes (Birt et al. 1997; Gunawan et al. 2020; Gregorin and Zanatta 2021). One of these, known as horny papillae (also called giant papillae), has been previously described in the family Phyllostomidae in species such as Carollia perspicillata, Glossophaga soricina (Masuko et al. 2007), Brachyphylla cavernarum, Phyllonycteris poeyi, Artibeus jamaicensis, Leptonycteris sanborni, Leptonycteris nivalis, Micronycteris megalotis, and Mimon bennettii (Griffiths 1982), as well as in the families Mormoopidae and Noctilionidae in Pteronotus rubiginosus and Noctilio albiventris, respectively (Gregorin and Zanatta 2021). This type of papillae, together with the filiform papillae, are mechanical structures whose microstructures and processes are fundamental for adhesion, perforation, and fixation of food during flight (Birt et al. 1997; Abumandour and El-Bakary 2013). For this reason, their abundance and distribution between the apex and the body, along with their directionality, facilitate the movement of liquids and the fibers of fleshy fruits towards the pharynx (Birt et al. 1997; Mqokeli and Downs 2013).
The giant bicuspid filiform papillae are characterized by their large size and have previously been described only in Artibeus jamaicensis (Griffiths 1982). However, this term has also been used inconsistently to refer to horny papillae, although the two do not represent the same morphological type (Elizalde-Arellano et al. 2004a, b; Mqokeli and Downs 2013). The keratinization, abundance, and size of these papillae play essential roles in swallowing and food transport by squeezing and crushing fruits against the palate (Birt et al. 1997; Mqokeli and Downs 2013).
Circumvallate papillae, on the other hand, are characterized by the presence of taste buds on both their surface and sides (Greenbaum and Phillips 1974). Abumandour and El-Bakary (2013) suggested that an increase in the number of circumvallate papillae enhances gustatory perception by broadening the sense of taste, thereby increasing the sensitivity and detection of food in the root region, while a greater number of fungiform papillae improves the perception of food before it progresses through the digestive system (Ojima et al. 2000; Ojima 2001).
The presence of four circumvallate papillae—two medial, one right lateral, and one left lateral—has also been reported for species of Carollia perspicillata, Glossophaga soricina, Leptonycteris yerbabuenae and L. nivalis of the family Phyllostomidae. In the family Molossidae, one pair of circumvallate papillae has been described (Abumandour and El-Bakary 2013), while in other families of aerial insectivores of the Neotropics, both one-pair and two-pair patterns have been documented (Gregorin and Zanatta 2021).
On the other hand, in the family Pteropodidae (Hutcheon and Kirsch 2006) there are species such as Rousettus aegyptiacus, R. amplexicaudatus, and Cynopterus brachyotis are recognized as frugivores but present three circumvallate papillae in a triangular pattern (Greenbaum and Phillips 1974; Gregorin 2003; Jackowiak et al. 2009; Abumandour and El-Bakary 2013).
Frugivorous and nectarivorous species tend to exhibit a greater number of circumvallate papillae. In addition, a pattern is observed where the family Pteropodidae generally has three circumvallate papillae, while families such as Phyllostomidae, Molossidae, Noctilionidae, Emballonuri-dae, Furipteridae, Thyropteridae, Mormoopidae, Natalidae and Vespertilionidae display greater variation in papillae number (Masuko et al. 2007; Gregorin and Zanatta 2021; Gregorin 2003), even reaching zero as in the case of Desmodus rotundus (Masuko et al. 2007). Probably species with frugivorous feeding habits possess a greater number of circumvallate papillae due to the need to determine the quality of food prior to ingestion.
The horny papillae are typically associated with the frugivorous habit; however, their presence has also been reported in specimens with insectivorous and piscivorous diets (Elizalde-Arellano et al. 2004a, b; Gregorin and Zanatta 2021). Similarly, giant bicuspid papillae, although recorded only in one frugivorous species, are thought to occur in other members of the family Phyllostomidae (Griffiths 1982). The location, shape, and arrangement of the mechanical papillae may be associated with the compensation for missing teeth across bat families, since their dental formulas vary considerably. These papillae are therefore essential for the adhesion and fixation of food during flight, a role fulfilled through the microstructures and processes present in the lingual papillae (Mqokeli and Downs 2013; Díaz et al. 2021).
This description links the morphology of the lingual papillae, their microstructures, and arrangement with frugivorous trophic habits, given that the topography and types of mechanical papillae serve critical roles. In addition, the taste buds enhance the sensation and perception of fruit (Birt et al. 1997; Mqokeli and Downs 2013; Gunawan et al. 2020). For P. infuscus, there are records of fruit consumption from the families Moraceae and Solanaceae (Arias and Pacheco 2019). Based on the lingual morphological description, it also shares papillary types such as bifid, tricuspid, and horny papillae with bats of the insectivorous guild; this could indicate an occasional consumption of insects.
Several studies have shown that morphological changes can occur in the lingual papillae at the intraspecific level or even between subspecies, driven by the availability of resources in the environments they inhabit. Thus, individuals may consume one resource more frequently than another, and this pattern, when associated with geographical distribution, can influence tongue morphology, as described in R. aegyptiacus (Mohamed et al. 2013). Differences related to life stage have also been reported: while certain papillae, such as taste buds, are fully developed at birth, others, such as filiform papillae, are underdeveloped in newborns and only reach full maturation at later stages (Trzcielińska-Lorych et al. 2009). For this order, it has been suggested that adaptations in the lingual surface morphology—and specifically in papillary typology—are correlated with trophic habits and may even serve as valid taxonomic characters (Harper et al. 2013; Abumandour and El-Bakary 2013; Gregorin and Zanatta 2021; Attaallah et al. 2023).
Eleven types of papillae were identified on the lingual surface of P. infuscus. In the root region, circumvallate papillae were identified, along with circular fungiform papillae and conical papillae, the latter two being exclusive to the root and the transitional zone with the body. In the lingual body, giant bicuspid papillae predominated, and to a lesser extent tricuspid filiform papillae accompanied by triangular fungiform papillae. Along the edges of the tongue, giant bicuspid papillae decreased in size, while fungiform papillae were identified. The most abundant papillae in the lingual body were strictly filiform papillae, extending from the terminal region of the middle body to the apex of the tongue. The greatest diversity of papillae was observed at the apex, with an island of horny papillae surrounded by digitiform papillae and bifid filiform papillae, all of which were directed toward the central canal of the tongue and pharynx.
To continue this type of study, it is necessary to establish a clear and comprehensive nomenclature that provides a classification of papillae and facilitates their identification, since several synonymies and irregular descriptions are currently present for the different papillary types, hindering accurate description and subsequent data standardization.
Finally, it is necessary to increase the number of individuals studied within the same species to confirm that the description effectively corresponds to the species. A broader sample size could reveal intraspecific morphological variation associated with factors such as age, sex, and geographical distribution. These sources of variation can significantly influence the interpretation of lingual traits and should therefore be carefully considered when establishing diagnostic characters at the species level.
Acknowledgments
The authors would like to thank D Otalora of the Scanning Electron Microscopy Laboratory of the Pontificia Universidad Javeriana and D Corredor of the Corporación Universitaria Minuto de Dios for their support in capturing the images and their subsequent analysis, and the MHNUD for supplying the sample. Finally, thanks to the Universidad Distrital Francisco José de Caldas for the academic education provided during these years.
Literature cited
Abumandour, M. M., and R. M. El-Bakary. 2013. Morphological and scanning electron microscopic studies of the tongue of the Egyptian fruit bat (Rousettus aegyptiacus) and their lingual adaptation for its feeding habits. Veterinary Research Communications 37:229–238.
Arias, E., and V. Pacheco. 2019. Dieta y estructura trófica de un ensamblaje de murciélagos en los bosques montanos del Santuario Nacional Pampa Hermosa, Junín, Perú. Revista Peruana de Biología 26:169–182.
Attaallah, A., et al. 2023. Comparative studies on the tongue of the Egyptian fruit bat (Rousettus aegyptiacus) and the common quail (Coturnix coturnix). Egyptian Journal of Basic and Applied Sciences 10:476–492.
Birt, P., L. S. Hall, and G. C. Smith. 1997. Ecomorphology of the tongues of Australian megachiroptera (Chiroptera: Pteropodidae). Australian Journal of Zoology 45:369–384.
Cerón-Hernández, J. A., et al. 2022. Diversidad, tipos de dieta de murciélagos y su respuesta a bordes de bosque mesófilo de montaña, Veracruz, México. Ecosistemas y Recursos Agropecuarios 9:e3110.
Calonge, B. H. 2009. Dieta y estructura trófica del ensamblaje de murciélagos en un sistema de ganadería extensiva en remanentes de bosque seco tropical en Córdoba (Colombia). Pontificia Universidad Javeriana, Bogotá, Colombia.
Díaz, M., et al. 2021. Clave de identificación de los murciélagos neotropicales. Programa de Conservación de los Murciélagos de Argentina, Buenos Aires, Argentina.
Doran, G. 1975. Review of the evolution and phylogeny of the mammalian tongue. Acta Anatomica 91:118–129.
Dumont, E., et al. 2012. Morphological innovation, diversification and invasion of a new adaptive zone. Proceedings of the Royal Society B: Biological Sciences 279:1797–1805.
Elizalde-Arellano, C., E. Uría-Galicia, and J. C. López-Vidal. 2004a. Morfología lingual del murciélago piscívoro Noctilio leporinus (Chiroptera: Noctilionidae). Acta Zoológica Mexicana 20:69–78.
Elizalde-Arellano, C., E. Uría-Galicia, and J. C. López-Vidal. 2004b. Estructura anatómica e histológica de la lengua del murciélago frugívoro Centurio senex (Chiroptera: Phyllostomidae). Acta Zoológica Mexicana 20:31–37.
Fleming, T. H., and N. Muchhala. 2008. Nectar‐feeding bird and bat niches in two worlds: pantropical comparisons of vertebrate pollination systems. Journal of Biogeography 35:764–780.
Gardner, A. L. (ed.). 2008. Mammals of South America: Marsupials, Xenarthrans, Shrews, and Bats, vol. 1. University of Chicago Press, Chicago, EE. UU.
González-Terrazas, T. P., et al. 2012. Morphological specialization influences nectar extraction efficiency of sympatric nectar-feeding bats. Journal of Experimental Biology 215:3989–3996.
Greenbaum, I. F., and C. J. Phillips. 1974. Comparative anatomy and general histology of tongues of long-nosed bats (Leptonycteris sanborni and L. nivalis) with reference to infestation of oral mites. Journal of Mammalogy 55:489–504.
Gregorin, R., and P. A. Zanatta. 2021. Comparative morphology of tongue surface in Neotropical aerial insectivore bats (Mammalia: Chiroptera). Papéis Avulsos de Zoologia 61:e20216186.
Gregorin, R. 2003. Comparative morphology of the tongue in free-tailed bats (Chiroptera, Molossidae). Iheringia. Série Zoologia 93:213–221.
Griffiths, T. A. 1982. Systematics of the New World nectar feeding bats (Mammalia: Phyllostomidae) based on morphology of the hyoid and lingual regions. American Museum Novitates 2742:1–45.
Gunawan, G., et al. 2020. Morphological study of the lingual papillae in the fruit bat (Rousettus amplexicaudatus) by scanning electron microscopy and light microscopy. Anatomia, Histologia, Embryologia 49:173–183.
Harper, C. J., S. M. Swartz, and E. L. Brainerd. 2013. Specialized bat tongue is a hemodynamic nectar mop. Proceedings of the National Academy of Sciences 110:8852–8857.
Hill, J. E., and J. D. Smith. 1984. Bats: A Natural History. University of Texas Press, Austin, EE. UU.
Hutcheon, J. M., and J. A. Kirsch. 2006. A moveable face: deconstructing the Microchiroptera and a new classification of extant bats. Acta Chiropterologica 8:1–10.
Iwasaki, S. 2022. Evolution of the structure and function of the vertebrate tongue. Journal of Anatomy 201:1–13.
Jackowiak, H., J. Trzcielińska-Lorych, and S. Godynicki. 2009. The microstructure of lingual papillae in the Egyptian fruit bat (Rousettus aegyptiacus) as observed by light microscopy and scanning electron microscopy. Archives of Histology and Cytology 72(1).
Lee, W. J., et al. 2017. Tongue-driven sonar beam steering by a lingual-echolocating fruit bat. PLoS Biology 15:e2003148.
Masuko, T. S., et al. 2007. Comparative scanning electron microscopic study of the lingual papillae in three species of bats (Carollia perspicillata, Glossophaga soricina and Desmodus rotundus). Microscopy and Microanalysis 13:280–281.
Mqokeli, B. R., and C. T. Downs. 2013. Palatal and lingual adaptations for frugivory and nectarivory in the Wahlberg’s epauletted fruit bat (Epomophorus wahlbergi). Zoomorphology 132:111–119.
Ojima, K., et al. 2000. Angio architectural form, functional distributive pattern and classification of the filiform papillae on the crossbred Japanese cat tongue anterodorsal surface in scanning electron microscopic specimens. Annals of Anatomy - Anatomischer Anzeiger 182:47–52.
Ojima, K. 2001. Functional role and angioarchitectural arrangement of the filiform and fungiform papillae on the medial-dorsal surface of the beagle dog tongue. Annals of Anatomy 183:325–329.
Ramírez-Chaves, H. E., et al. 2024. Mamíferos de Colombia. v1.14. Sociedad Colombiana de Mastozoología, Bogotá, Colombia.
Roberts, L. H. 1975. Confirmation of the echolocation pulse production mechanism of Rousettus. Journal of Mammalogy 56:218–220.
Rodríguez-Bolaños, A. 2020. Colección de Mamíferos, Museo de Historia Natural de la Universidad Distrital Francisco José de Caldas (MHNUD-M). Mammalogy Notes 6:0120.
Rojas, D. Á., et al. 2011. When did plants become important to leaf-nosed bats? Diversification of feeding habits in the family Phyllostomidae. Molecular Ecology 20:2217–2228.
Shanahan, M., et al. 2001. Fig-eating by vertebrate frugivores: a global review. Biological Reviews, 76:529–572.
Sonntag, C. F. 1926. The comparative anatomy of the tongues of the Mammalia. XII. Summary, classification and phylogeny. Journal of Mammalogy 7:701–762.
Trzcielińska-Lorych, J. H., et al. 2009. Morphology and morphometry of lingual papillae in adult and newborn egyptian fruit bats (Rousettus aegyptiacus). Journal of Veterinary Medicine Series C: Anatomia Histologia Embryologia 38:370–376.
Weinberg, M., Y. Nissan, and Y. Yovel. 2023. Egyptian Fruit Bat Rousettus aegyptiacus (Geoffroy, 1810). Pp. 1–26 in Handbook of the Mammals of Europe. Springer International Publishing, Cham, Switzerland.
Winter, Y., and Von Helversen, O. 2003. Operational tongue length in phyllostomid nectar-feeding bats. Journal of Mammalogy 84: 886–896.
Associate editor: Beatríz del S. Bolívar Cimé
Submitted: June 30, 2025; Reviewed: January 28, 2026
Accepted: February 12, 2026; Published on line: April 14, 2026

Figure 1. Dorsal view of the tongue of P. infuscus and distribution of the diverse types of papillae in the root , body, and apex regions; (cv) circumvallate papillae, (fu) fungiform papillae, triangular fungiform papilla (fn), (cp) conical papillae, (fb) giant bicuspid papillae, (ft) tricuspid papillae, and (hp) horny papillae.
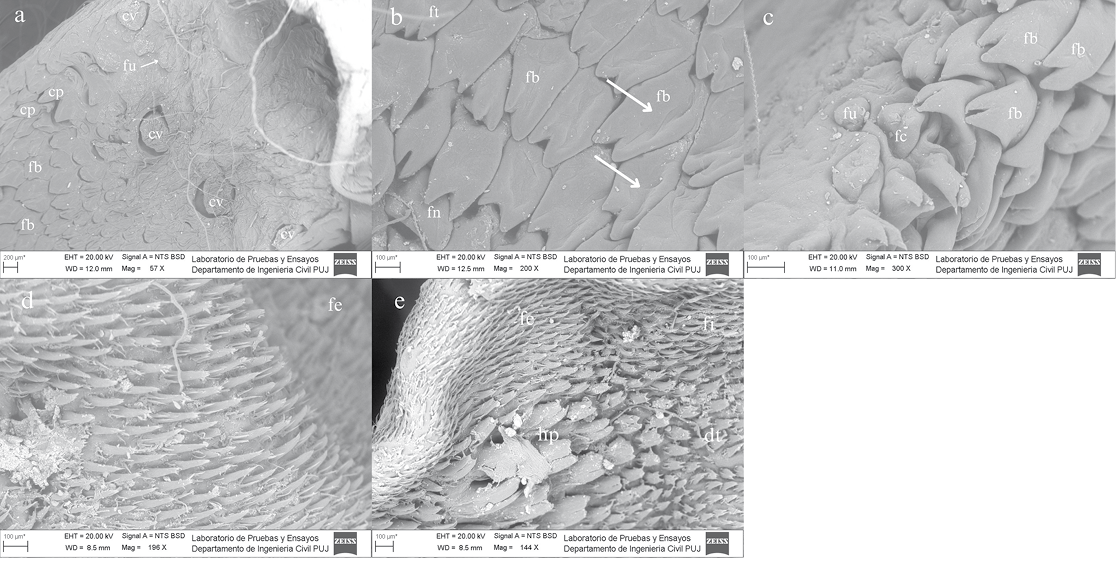
Figure 2. Scanning electron micrographs (SEM) of the tongue of Platyrrhinus infuscus showing the distribution and morphology of the different types of lingual papillae. a) Dorsal view of the central and lateral root region: (cv) circumvallate papillae, (fu) circular fungiform papillae, (cp) conical papillae, (fb) giant bicuspid filiform papillae. b) Mid-body region, dorsal view: (fb) giant bicuspid filiform papillae, (ft) tricuspid filiform papillae, (fn) triangular fungiform papilla, central sulci (white arrows). c) Lingual margin of the body near the root, right lateral view: (fc) vaulted fungiform papillae, (fu) circular fungiform papilla, (fb) giant bicuspid filiform papillae. d) Body–apex transitional region, dorsal view: (fe) strictly filiform papillae. e) Distal apex, dorsal view: (fe) strictly filiform papillae, (dt) finger-like papillae, (fi) bifid filiform papillae, (hp) horny papillae. High-vacuum SEM was performed at 20 kV.