THERYA NOTES 2026, Vol. 7:89-95
Turgida turgida (Nematoda: Physalopteridae) parasitizing
Water Oposssum (Chironectes minimus Zimmermann, 1780)
in the Western Cordillera of Colombia and updated
checklist of its associated parasites
Turgida turgida (Nematoda: Physalopteridae) parasitando zarigüeya acuática (Chironectes minimus Zimmermann, 1780) en la Cordillera Occidental de Colombia y lista actualizada de sus parásitos asociados
Diego Álvarez-Arellano1,2*, José Leandro Pérez-Bedoya3, John Harold Castaño2,
Gerardo Elejalde2, Shirley Palacios-Castro1, and Manuela Londoño-Raigosa2
1Laboratorio de Entomología, Corporación Universitaria Santa Rosa de Cabal, Km 4 vía Chinchiná, Vereda El Jazmín, Santa Rosa de Cabal, Risaralda, Colombia. E-mail: diego.alvarez@unisarc.edu.co (DÁA); shirley.palacios@unisarc.edu.co (SPC).
2Grupo de Investigación en Biología de la Conservación y Biotecnología, Corporación Universitaria de Santa Rosa de Cabal, Km 4 vía Chinchiná, Vereda El Jazmín, Santa Rosa de Cabal, Risaralda, Colombia. E-mail: john.castano@unisarc.edu.co (JHC); gerardo.elejalde@unisarc.edu.co (GE); manuela.londonor@unisarc.edu.co (MLR).
3Institución Universitaria Visión de las Américas, Pereira, Risaralda, Colombia. E-mail: jose.perez@unisarc.edu.co (JLPB).
*Corresponding author
Chironectes minimus is the only marsupial species of a semi-aquatic nature and is widely distributed in tropical and subtropical habitats from Central to South America. Although it has a wide distribution, little is known about aspects of its biology, as well as its parasites because it is considered a difficult species to sample. In this study, we report for the first time the presence of the nematode Turgida turgida in C. minimus in an area of the Western Cordillera in Colombia and compile a list of its other associated parasites. While preparing a specimen of C. minimus to enter the Mammal Collection of the Corporación Universitaria Santa Rosa de Cabal (CUS-M), six individuals of T. turgida were found when their stomach contents were checked. For scanning electron microscope analysis (SEM), all the samples were cleaned in saline solution and mounted on supports using carbon adhesive tape, coated with gold by sputtering in a cathodic sputtering machine. The nematodes found in the stomach of C. minimus were identified as Turgida turgida and correspond to five males and one female with a mean length of 2.45 cm and 4.59 cm, respectively. SEM revealed structures that characterize this species, such as ventrocaudal ornamentation patterns, and the presence of the 22nd broad, truncated caudal papilla on the male tail. We emphasize the importance of biological collections and recommend closer examination of specimens during the collection process, given the possibility of gathering relevant information on specimen-associated parasites, which could provide valuable insights into host-parasite relationships, ecological interactions and potential disease transmission pathways, ultimately allowing us to understand their implications for both public health and animal health.
Key words: Cloud forests; biological collections; neotropical marsupial; parasitic helminths; scanning electron microscope.
Chironectes minimus es la única especie de marsupial de carácter semiacuático y se encuentra ampliamente distribuida en hábitats tropicales y subtropicales desde Centroamérica hasta Sudamérica. A pesar de su amplia distribución, poco se conoce sobre aspectos de su biología, así como de sus parásitos debido a que es considerada una especie difícil de muestrear. En este estudio reportamos por primera vez la presencia del nematodo Turgida turgida en C. minimus en un área de la Cordillera Occidental en Colombia y compilamos una lista de sus otros parásitos asociados. Mientras se preparaba un espécimen de C. minimus para ingresar a la Colección de Mamíferos de la Corporación Universitaria Santa Rosa de Cabal (CUS-M), se encontraron seis individuos de T. turgida al revisar su contenido estomacal. Para el análisis de microscopía electrónica de barrido (MEB), todas las muestras fueron limpiadas en solución salina y montadas sobre soportes utilizando cinta adhesiva de carbono, recubiertas con oro por pulverización catódica. Los nematodos encontrados en el estómago de C. minimus fueron identificados como Turgida turgida y corresponden a cinco machos y una hembra con una longitud media de 2,45 cm y 4,59 cm, respectivamente. La MEB reveló estructuras que caracterizan a esta especie, como los patrones de ornamentación ventrocaudal y la presencia de la 22 papila caudal ancha y truncada en la cola del macho. Destacamos la importancia de las colecciones biológicas y recomendamos un examen más minucioso de los especímenes durante el proceso de recogida, dada la posibilidad de recopilar información relevante sobre los parásitos asociados a los especímenes, lo que podría aportar información valiosa sobre las relaciones huésped-parásito, las interacciones ecológicas y las posibles vías de transmisión de enfermedades, permitiéndonos en última instancia comprender sus implicaciones tanto para la salud pública como para la sanidad animal.
Palabras clave: Bosque de niebla; colecciones biológicas; helmintos parásitos; marsupial neotropical; microscopía electrónica de barrido.
© 2026 Asociación Mexicana de Mastozoología, www.mastozoologiamexicana.org
DOI: 10.12933/therya_notes-25-236
ISSN 2954-3614

The Water Opossum, Chironectes minimus Zimmermann, 1780 (Mammalia: Didelphidae) is the only marsupial species of a semi-aquatic nature and is widely distributed in tropical and subtropical habitats from Central America to South America (Ardente et al. 2013). It is mainly associated with riverbeds with stony substrates, clear water, and preserved riparian vegetation (Prieto-Torres and Pinilla-Buitrago, 2017). Although it has a wide distribution, little is known about aspects of its biology, as well as its parasites (Fernandez et al. 2015) because it is considered a difficult species to sample given that it is not attracted by conventional baits and because it mainly inhabits water sources, which makes it difficult to set traps (Bressiani and Graipel 2008). Parasites are essential in ecosystems because they participate in the regulation and structuring of the host population, intervening in processes such as competition, migration, reproduction and speciation (Bennati et al. 2023), and both biotic and abiotic factors influence their interaction with hosts (Cirino et al. 2020).
The literature documents that parasitic helminths in wildlife are rare and there are few reviews of them (Beveridge and Spratt 2015). This may be due to the fact that many species are cryptic and require molecular techniques for identification, and to the scarce availability of active taxonomists who can determine them by morphological characteristics (Kim and Byrne 2006). This results in the fact that many helminth species deposited in biological collections have not yet been described and numerous marsupial species have not been examined for parasites (Beveridge and Gasser 2014), making it difficult to understand the biology and interaction between the two groups. The family Physalopteridae (Nematoda: Spiruroidea) comprises 22 genera and around 314 valid species (Hodda 2022), among which Turgida turgida Rudolphi, 1819 stands out as it is widely recorded in marsupials such as Didelphis aurita (Gomes et al. 2003), D. virginiana (Gray and Anderson 1982), among others. In this study, we report for the first time the presence of T. turgida in C. minimus, confirmed by scanning electron microscopy (SEM) in an area of the Western Cordillera in Colombia and compile a list of its other associated parasites.
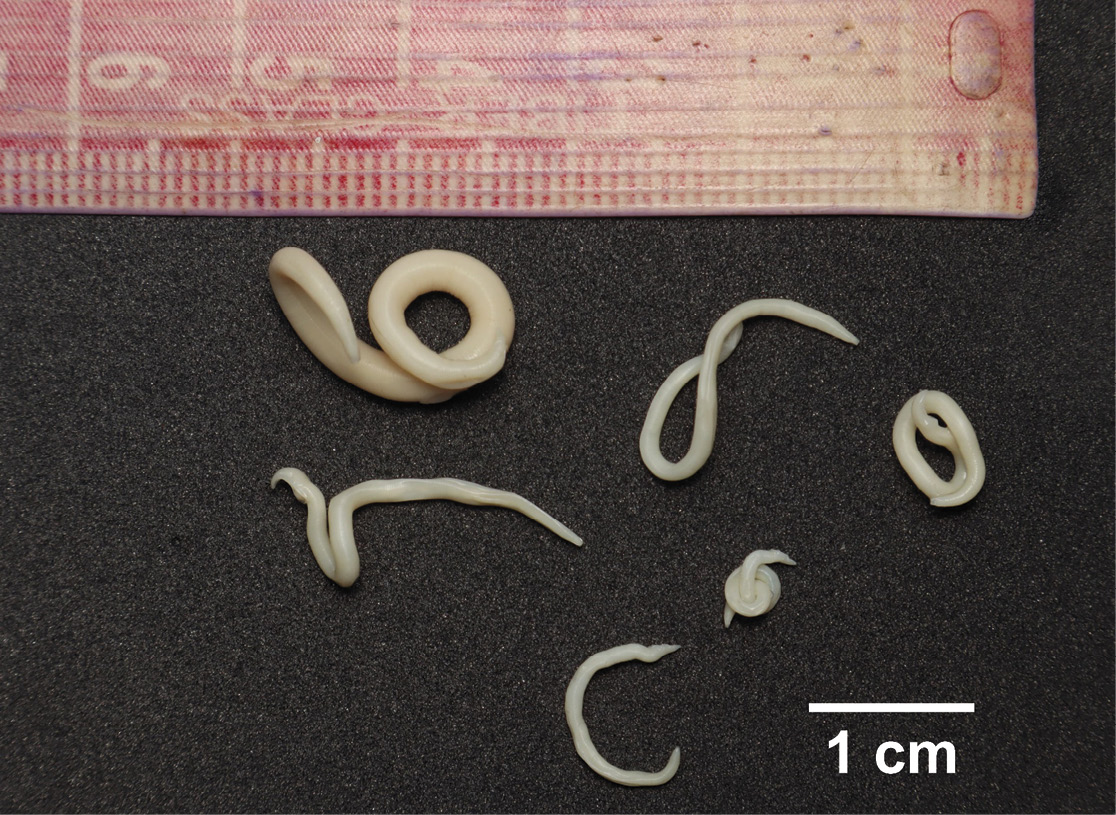
While preparing a specimen of C. minimus to enter the Mammal Collection of the Corporación Universitaria Santa Rosa de Cabal (CUS-M), six individuals of T. turgida were found when their stomach contents were checked (Figure 1). The specimen, a male C. minimus from the municipality of Pueblo Rico, Risaralda, Colombia (5° 16’ 21.0” N, 76° 1’ 36.0” W; 1169 m), was collected under the Framework Permit for the collection of wildlife specimens for scientific research purposes, Res 2004/2016 granted by the Corporación Autónoma Regional de Risaralda (CARDER), then was catalogued with the code CUS-M 0514; and samples of its parasitic helminths were preserved and fixed in a solution of 70 % alcohol and 5 % formaldehyde. For scanning electron microscopy analysis, the samples were cleaned in saline solution and mounted on supports using carbon adhesive tape, coated with gold by sputtering in a cathodic sputtering machine (Quorum model SC7620) and examined with a scanning electron microscope (Fei Quanta 250) at 12.5kV at the Laboratorio de Microscopía Electrónica of the Universidad de Caldas. Subsequently, the works of Matey et al. (2001), Chaubad (2009) and Humberg et al. (2011) were used for taxonomic identification.
A literature review was conducted to compile the spe-cies of parasites reported in C. minimus, and the information is drawn from nine scientific articles retrieved from the Dialnet, Elsevier, Google Scholar, PubMed, Redalyc, and SciELO databases using the following keywords: arthro-pods, Chironectes minimus, Didelphidae, Didelphimorphia, helminths, marsupials, nematodes, parasites, hosts, Phy-salopteridae, Turgida turgida. The records were obtained from papers that addressed aspects of the natural history of the species (Marshall 1978; Smith 2009) and checklists or reports of specific parasite groups (Miyazaki et al. 1974;
Pinto et al. 2011; Lindardi 2012; Chero et al. 2017; Noronha et al. 2002; Varella et al. 2022; Jiménez et al. 2024).
The nematodes found in the stomach of C. minimus were identified as Turgida turgida and correspond to males (n = 5) and one female (n = 1), with a mean length of 2.45 cm (2.90; 3.18; 2.42; 1.85; 1.88 cm) and 4.59 cm, respectively (Figure 1). No other parasites were found in the specimen. SEM revealed structures that differentiate T. turgida from other Physalopteridae species, such as ventrocaudal ornamentation patterns (Figure 2a, Figure 2b, Figure 2c), and the presence of the 22nd broad, truncated caudal papilla on the male tail (Figure 2d). The specimen examined had no apparent lesions on the stomach.
According to the literature review, C. minimus has been reported to have 18 parasites in different areas of the body (Table 1). Most of them are helminths, with 14 species (77.78 %) predominantly belonging to the genera Aspidodera and Cruzia, and the remaining four are arthropods. This distribution highlights a marked predominance of helminth parasites in C. minimus, suggesting that gastrointestinal and other internal parasitic infections constitute the most commonly documented host-parasite associations in this species. In contrast, arthropod parasites account for 22.22 % of the total diversity of parasites documented in the literature. Records indicate that helminths are found in internal anatomical regions, particularly associated with the gastrointestinal tract, while arthropods are mainly associated with external regions of the body.
This data corresponds to information collected over more than 20 years, most of which comes from occasional records. They appear in brief mentions in studies on specific parasites and host lists and are often part of supplementary information when the main subject of the study is on other species of Didelphimorphia. Although not all papers specify the location where the parasite and host were recorded, there is information on the parasites Rhopalias caballeroi, Spirura guianensis and Travassostrongylus callis in Bolivia, Adoratopsylla antiquorum, Aspidodera raillieti, Cruzia tentaculata, Dipetalonema sp., Litomosoides sp., Trichuris minuta and Turgida turgida in Brazil; and Paragonimus amazonicus in Peru.
Turgida turgida is a parasite reported in several species of marsupials such as Didelphis albiventris, D. aurita, D. marsupialis, D. virginiana and Philander opossum, being present from North America to South America and normally associated with the greater curvature of the stomach (Gray and Anderson 1982). In the family Physalopteridae the species appear to be selective to the site of infection, differing from the genus Physaloptera, which can infect the lesser covering of the stomach, the pylorus, or the fundic region (Humberg et al. 2011). These parasitic helminths can cause gastric perforations and sepsis, thus Nichelason et al. (2008) highlight T. turgida as a species with the ability to significantly influence morbidity and mortality in didelphids. Its life cycle is indirect, beginning when an insect ingests the parasite’s eggs, whose larvae subsequently become encysted in the external wall of the intestine (Anderson 2000), the definitive host consumes the insect and then the nematode matures sexually and begins to produce eggs that will be released into the environment through the feces, thus completing the cycle (Gray and Anderson 1982). As intermediate hosts, the orders Blattodea, Coleoptera, and Orthoptera have been reported (Zago-Filho 1958; Anderson 2000). On the other hand, paratenic hosts such as amphibians and reptiles have also been reported (Widmer 1970; Humberg et al. 2011). Chironectes minimus has mainly a carnivorous diet, frequently consuming different aquatic organisms such as fish, crustaceans, frogs and insects (Hume 2005), therefore, it is possible that in the case of T. turgida the form of infection towards C. minimus is through its diet, given the wide diversity in its diet which could be a factor for this species to be more vulnerable to prey that are intermediate hosts, thus enhancing the spectrum of infections (Álvarez-Coto 2018), in the same way that happens in other didelphids (Gomes et al. 2003). Previously, Noronha et al. (2002) and Pinto et al. (2011) report T. turgida as a stomach parasite of C. minimus in Brazil in State of Rio de Janeiro, municipality of Santo Antônio, State of Pará. In addition, there are records of arthropods, cestodes, trematodes, and other nematodes parasitizing different areas of the body (Table 1). According to the literature, T. turgida has high prevalence rates in other Didelphimorphia (Table 2) in countries such as Bolivia (Jiménez et al. 2024), Brazil (Silva and Costa 1999; Boullosa et al. 2017; Costa-Neto et al. 2019; Freitas et al. 2022), Mexico (Cañeda-Guzmán 1997; Monet-Mendoza et al. 2005), and the United States (Gray and Anderson 1982). This high prevalence may be related to the ecological and trophic characteristics of these marsupials, which could increase their exposure to infectious stages present in the environment or in intermediate hosts. It may also suggest that this nematode is well adapted to these hosts and that its transmission is relatively efficient, playing a significant role within the parasitic community associated with them (Freitas et al. 2022). From a population perspective, infections with high prevalence could influence the physiological condition of individuals, especially when parasite loads are high, which could potentially affect their survival or reproductive success (Boullosa et al. 2017). The higher number of T. turgida records in countries such as Bolivia, Brazil, Mexico and the United States does not necessarily reflect its actual distribution on the continent, but rather differences in sampling effort and the intensity of parasitological studies conducted in these areas. In this regard, the absence of records in other countries of the Americas could be associated with a smaller number of studies focused on the parasitic fauna of Didelphimorphia, rather than a true absence of the nematode.
Spratt and Beveridge (2016) estimate that about 26 % of the global marsupial fauna has not yet been examined for parasites. The lack of representativeness of this group in some biological collections may be because they are rare or threatened, limiting the opportunity to collect parasites from them (Beveridge and Spratt 2015). Although the diversity of helminths and other parasites is well documented in Australian marsupials (Beveridge and Gasser 2014; Beveridge and Spratt 2015; Spratt and Beveridge 2016), in contrast, for the Neotropics, having fewer species, efforts to know the parasites of marsupials have been focused mainly on the genus Didelphis (Jiménez et al. 2011; Acosta-Virgen et al. 2015; Freitas et al. 2022) because these species inhabit peri-urban areas, in addition to being compatible hosts and reservoirs of diseases that affect humans (Bezerra-Santos et al. 2021). Previous studies suggest that the family Didelphidae are frequent hosts of helminths (Benatti et al. 2023).
Chironectes minimus has been considered a rare or infrequent species, however, the capture method proposed by Bressiani and Graipel (2008) demonstrates that it may be more common than previously thought; representing an opportunity to better understand the parasites associated with this species and other aspects of its biology. Considering that the last recorded occurrence of the helminth T. turgida in C. minimus was more than two decades ago, this record contributes to the knowledge of parasite-host relationships between these species, in addition to being the first record in Colombia. Furthermore, we emphasize the importance of biological collections and recommend further examination of specimens during the collection process, given the possibility of gathering relevant information regarding the parasites associated with the specimens, allowing us to understand their possible implications for public and animal health (Benatti et al. 2023).
Acknowledgements
We thank J. C. Cepeda-Duque, A. Parra and G. A. Bolaños Gallardo for their valuable support during the development of this work. This research was funded by the Corporación Universitaria Santa Rosa de Cabal.
Acosta-Virgen, K., et al. 2015. Helminths of three species of opossums (Mammalia, Didelphidae) from Mexico. ZooKeys 511:131–152.
Álvarez-Coto, A. 2018. Descripción taxonómica y parámetros ecológicos de Turgida turgida asociado al tlacuache (Didelphis virginiana) en la localidad La Comunidad, Jilotepec, Estado de México, (Bachelor Thesis). Universidad Nacional Autónoma de México. Estado de México.
Ardente, N., et al. 2013. Mammalia, Didelphimorphia, Didelphidae, Glironia venusta Thomas, 1912 and Chironectes minimus (Zimmermann, 1780): distribution extension for eastern Amazonia. Check List 9:1104–1107.
Álvarez-Coto, A. 2018. Descripción taxonómica y parámetros ecológicos de Turgida turgida asociado al tlacuache (Didelphis virginiana) en la localidad La Comunidad, Jilotepec, Estado de México, (Bachelor Thesis). Universidad Nacional Autónoma de México. Estado de México.
Ardente, N., et al. 2013. Mammalia, Didelphimorphia, Didelphidae, Glironia venusta Thomas, 1912 and Chironectes minimus (Zimmermann, 1780): distribution extension for eastern Amazonia. Check List 9:1104–1107.
Benatti, D., et al. 2023. Endoparasites of marsupials in fragments of the Atlantic rainforest, western Paraná State, Brazil. Revista Brasileira de Parasitologia Veterinária 32: e005823.
Beveridge, I. And Gasser, R. B. 2014. Diversity in parasitic helminths of Australasian marsupials and monotremes: a molecular perspective. International Journal for Parasitology 44:859–864.
Beveridge, I. And Spratt, D. M. 2015. Biodiversity and parasites of wildlife: helminths of Australasian marsupials. Trends in Parasitology 31:142–148.
Bezerra-Santos, M. A., et al. 2021. Didelphis spp. opossums and their parasites in the Americas: A One Health perspective. Parasitology Research 120:4091–4111.
Boullosa, R. G., et al. 2017. Ecological aspects of nematode parasites of Didelphis aurita (Didelphimorphia, Didelphidae) in urban-sylvatic habitats in Rio de Janeiro, Brazil. Oecologia Australis 21:54–61.
Bressiani, V. B. And Graipel, M. E. 2008. Comparação de métodos para captura da cuíca-d’água, Chironectes minimus (Zimmerman, 1780) (Mammalia, Didelphidae) no sul do Brasil. Mastozoología Neotropical 15:33–39.
Cañeda-Guzmán, I. C. 1997. Parásitos de tres especies de marsupiales de la estación Los Tuxtlas y algunas zonas cercanas, Veracruz, México. B. S. Thesis, Facultad de Ciencias, Universidad Nacional Autónoma de México, Mexico City.
Chabaud, A. G. 2009. Spirurida: Key to genera of the Physalopteroidea. Pp. 345–349 in Keys to the Nematode Parasites of Vertebrates (Anderson R. C., Chabaud A. G. and Willmott, S. eds.). Archival Volume. United Kingdom.
Chero, J. D., et al. 2017. Helminths of the common opossum Didelphis marsupialis (Didelphimorphia: Didelphidae), with a checklist of helminths parasitizing marsupials from Peru. Revista Mexicana de Biodiversidad 88:560–571.
Cirino, B. S., et al. 2020. First study on the helminth community structure of the neotropical marsupial Metachirus myosuros (Didelphimorphia, Didelphidae). Revista Brasileira de Parasitologia Veterinária 29: e005420.
Costa-Neto S. F, et al. 2019. Metacommunity structure of the helminths of the black-eared opossum Didelphis aurita in peri-urban, sylvatic and rural environments in south-eastern Brazil. Journal of Helminthology 93:720–731.
Fernandez, F. A., et al. 2015. Natural history of the water opossum Chironectes minimus: a review. Oecologia Australis 19:47–62.
Freitas, L. D. C., et al. 2022. Helminth community structure of Didelphis marsupialis (Didelphimorphia, Didelphidae) in a transition area between the Brazilian Amazon and the Cerrado. Revista Brasileira de Parasitologia Veterinária 31: e002922.
Gomes, D. C., et al. 2003. Nematode parasites of marsupials and small rodents from the Brazilian Atlantic Forest in the State of Rio de Janeiro, Brazil. Revista Brasileira de Zoologia 20:699–707.
Gray, J. B. and Anderson, R. C. 1982. Observations on Turgida turgida (Rudolphi, 1819) (Nematoda: Physalopteroidea) in the American opossum (Didelphis virginiana). Journal of Wildlife Diseases 18:279–285.
Hodda, M. 2022. Phylum Nematoda: a classification, catalogue and index of valid genera, with a census of valid species. Zootaxa 5114:1–289.
Humberg, R. M., et al. 2011. Turgida turgida (Nematoda: Physalopteridae) parasitic in white-bellied opossum, Didelphis albiventris (Marsupialia: Didelphidae), state of Mato Grosso do Sul, Brazil. Pesquisa Veterinária Brasileira 31:78–80.
Hume, I. D. 2005. Nutrition of marsupials in captivity. International Zoo Yearbook 39:117–132.
Jiménez, F. A., et al. 2024. Gastrointestinal helminths of opossums (Mammalia: Didelphidae) from Bolivia. Parasitology 151:637–649.
Jiménez, F. A., et al. 2011. Structure of parasite component communities of didelphid marsupials: insights from a comparative study. Journal of Parasitology 97:779–787.
Kim, K. C. and Byrne, L. B. 2006. Biodiversity loss and the taxonomic bottleneck: emerging biodiversity science. Ecological Research 21:794–810.
Linardi, P. M. 2012. Os ectoparasitos de marsupiais brasileiros. Pp. 127–155 in Os Marsupiais do Brasil: Biologia, Ecologia e Conservacao (Cáceres N.C., ed.). Federal University of Mato Grosso Press, Brazil.
Marshall, L. G. 1978. Chironectes minimus. Mammalian Species 109:1–6.
Miyazaki, I., et al. 1974. Mature Paragonimus amazonicus first found from a water opossum in Peru. The Journal of Parasitology 60:547–548.
Monet-Mendoza, A., et al. 2005. Helminths of the Virginia opossum Didelphis virginiana (Mammalia: Didelphidae) in Mexico. Journal of Parasitology 91:213–219.
Nichelason, A. E., et al. 2008. Evaluation of Cruzia americana, Turgida turgida, and Didelphostrongylus hayesi infection in the Virginia opossum (Didelphis virginiana) and risk factors along the California coast. Journal of Parasitology 94:1166–1168.
Noronha, D., et al. 2002. A survey of new host records for nematodes from mammals deposited in the Helminthological Collection of the Oswaldo Cruz Institute (CHIOC). Revista Brasileira de Zoologia 19:945–949.
Pinto, R. M., et al. 2011. Nematodes from mammals in Brazil: an updating. Neotropical Helminthology 5:139–183.
Silva, M. G. Q., and Costa, H. M. A. 1999. Helminths of White-bellied Opossum from Brazil. Journal of Wildlife Diseases 35:371–374.
Smith, P. 2009. Chironectes minimus. In: FAUNA Paraguay: Handbook of the Mammals of Paraguay, Volume 1: Marsupialia. www.faunaparaguay.com. Accessed on January 12, 2025.
Varella, K., et al. 2022. Population genetic structure and phenotypic diversity of Aspidodera raillieti (Nematoda: Heterakoidea), a parasite of Didelphini marsupials in Brazil’s South and Southeast Atlantic Forest. Parasites & Vectors 15:203.
Widmer, E. A. 1970. Development of third-stage Phy-saloptera larvae from Crotalus viridis Rafinesque, 1818 in cats with notes on pathology of the larvae in the reptile (Nematoda, Spiruroidea). Journal of Wildlife Diseases 6:89–93.
Zago-Filho, H. 1958. Contribuição para o conhecimento do ciclo evolutivo da Turgida turgida (Rud., 1819) Trav., 1920 (Nematoda: Spiruroidea). Arquivos de Zoologia 11:99–120.
Associate editor: Itandehui Hernández Aguilar
Submitted: May 13, 2025; Reviewed: March 18, 2026
Accepted: April 06, 2026; Published on line: April 23, 2026
Figure 1. Adult individuals of Turgida turgida found in the stomach wall of Chironectes minimus (CUS-M 0514) in Pueblo Rico, Risaralda, Colombia.
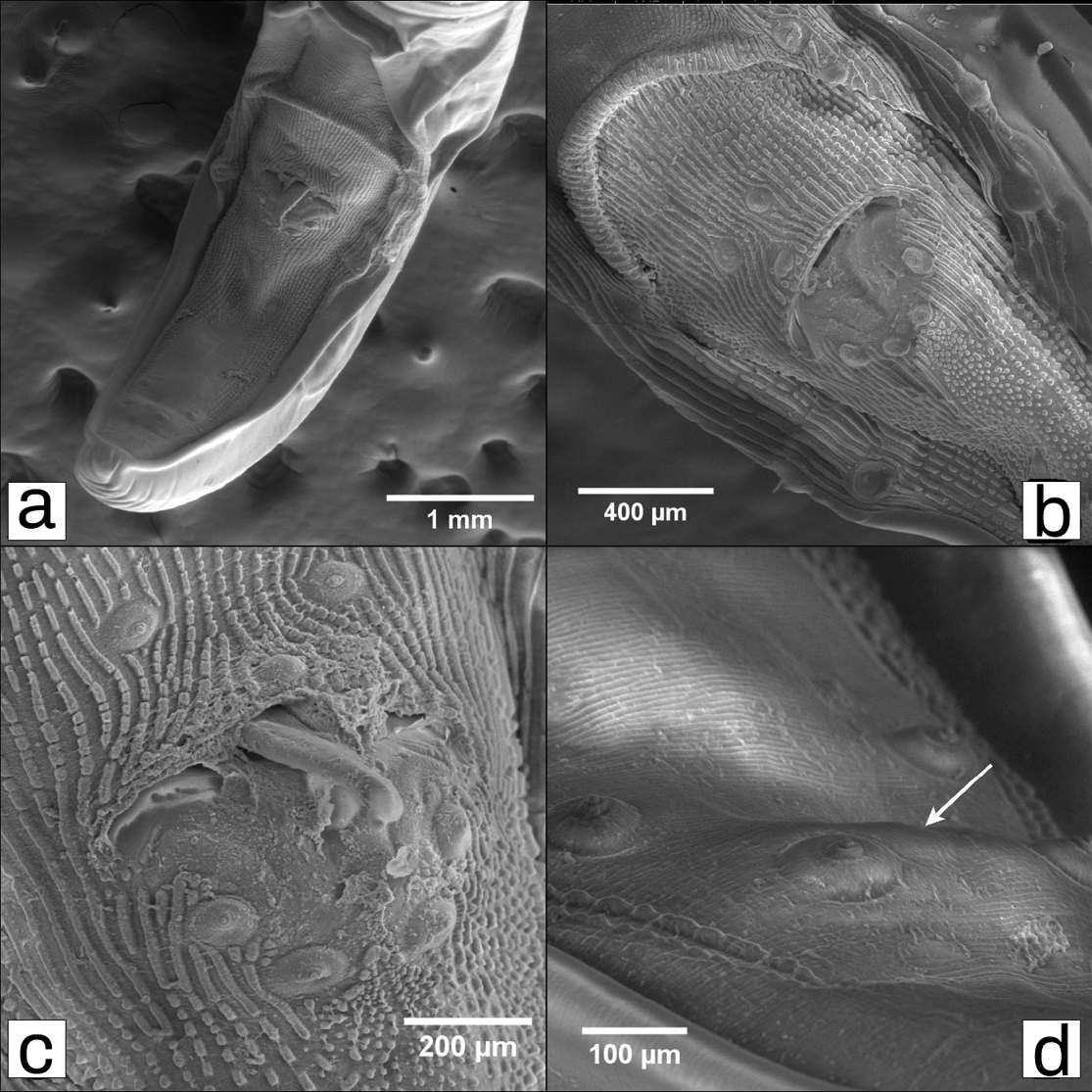
Figure 2. SEM micrographs of male specimens of Turgida turgida. (a) General view of the posterior end of the male, ventral side. The distribution of papillae can be seen, (b, c) Cloacal region, (D) Postcloacal papillae, including the broad, truncated papilla (white arrow).
Table 1. List of parasites found in Chironectes minimus.
|
Species/Taxon |
Infection site |
References |
|
Arthropoda |
||
|
Ctenophthalmidae |
||
|
Tegument |
||
|
Adoratopsylla sp. |
Tegument |
|
|
Doratopsylla sp. |
Tegument |
|
|
Trombiculidae |
||
|
Doloisia sp. |
Tegument |
|
|
Cestoda |
||
|
Diphyllobothriidae |
||
|
Ligula sp. |
Small intestine |
|
|
Sparganum sp. |
Muscle and subcutaneous tissue |
|
|
Nematoda |
||
|
Aspidoderidae |
||
|
Large intestine |
(Chero et al. 2017; Noronha et al. 2002, Pinto et al. 2011; Varella et al. 2022) |
|
|
Heligmosomidae |
||
|
Small intestine |
||
|
Kathlaniidae |
||
|
Cruzia tentaculata |
Excrements |
|
|
Onchocercidae |
||
|
Peritoneum |
||
|
Lungs |
||
|
Physalopteridae |
||
|
Turgida turgida |
Stomach |
(Noronha et al. 2002; Pinto et al. 2011; this work) |
|
Spiruridae |
||
|
Spirura guianensis |
Stomach |
|
|
Trichuridae |
||
|
Trichuris minuta |
Cecum |
|
|
Trematoda |
||
|
Opisthorchiidae |
||
|
Amphimerus sp. |
Bile ducts |
|
|
Paragonimidae |
||
|
Paragonimus amazonicus |
Lungs |
|
|
Rhopaliidae |
||
|
Rhopalias sp. |
Small intestine |
|
|
Rhopalias caballeroi |
Small intestine |
Table 2. Prevalence of Turgida turgida in other Didelphimorphia.
|
Species |
Prevalecence |
Country |
Reference |
|
Didelphis albiventris |
33% |
Bolivia |
|
|
73% |
Brazil |
||
|
Didelphis aurita |
70% |
Brazil |
|
|
75% |
Brazil |
||
|
Didelphis marsupialis |
75% |
Bolivia |
|
|
59% |
Brazil |
||
|
50% |
Mexico |
||
|
Didelphis virginiana |
50% |
Mexico |
|
|
60% |
Mexico |
||
|
72% |
United States |
||
|
Philander opossum |
9.5% |
Mexico |