Dasyprocta mexicana, commonly known as the Mexican agouti, is a rodent of the family Dasyproctidae, endemic to Mexico, whose distribution is restricted to the south-southeastern region of the country, encompassing the states of Veracruz, Oaxaca, Tabasco, and Chiapas. It has also been introduced to Cuba (Retana & Lorenzo 2002; Sánchez-Cordero et al. 2005; Borroto-Páez 2009; Santos-Moreno 2014; IUCN 2023). However, recent phylogenetic studies based on mitogenomic analyses have questioned the taxonomic validity of several species within the genus Dasyprocta, suggesting that some traditionally recognized species may represent lineages within broader species complexes. Consequently, the specific identity of D. mexicana requires molecular taxonomic revision (Ruiz-García et al. 2022). Morphologically, the species is characterized by a predominantly dark coloration, with adults weighing between 2 and 4 kg (Figure 1) (McWilliams 2009). This species has demonstrated notable adaptability to various habitat types, particularly tropical environments, where it primarily inhabits tall, medium, and lowland rainforests, as well as ecosystems with evergreen, semi-evergreen, and deciduous vegetation, including secondary growth and disturbed areas (Cortés-Marcial & Briones-Salas 2014; Zambrano et al. 2015; Hernández-Sánchez et al. 2017; Pozo-Montuy et al. 2019; Flores-Martínez et al. 2022; Franquesa-Soler et al. 2023). Dasyprocta mexicana exhibits frugivorous-granivorous feeding habits, playing a key eco-logical role as both seed disperser and seed predator in tropical ecosystems. It establishes interspecific competition for food and space with other large-bodied rodents, particularly the paca (Cuniculus paca) (Santos-Moreno & Pérez-Irineo 2013; Zambrano et al. 2015; Gallina & González-Romero 2018; Hernández-López & López-Castilla 2025).
The geographic distribution of D. mexicana is confined to tropical and subtropical regions of Mexico, especially in forested areas, where tropical forests provide essential habitat for its survival. Its strong association with tropical forests—such as Los Tuxtlas (Veracruz), the Selva Zoque in Oaxaca, and protected areas in Chiapas like the Selva El Ocote Biosphere Reserve—highlights its dependence on well-preserved ecosystems, even in areas subject to hunting pressure (Lira-Torres & Briones-Salas 2011; Zambrano et al. 2014; Ríos-Solís et al. 2021; Rodas-Trejo et al. 2025a). Nevertheless, there are also records of its use of degraded habitats, including oil palm plantations (Franquesa-Soler et al. 2023).
Dasyprocta mexicana is currently classified as Critically Endangered (CR) on the IUCN Red List (2023); however, it is not listed under any risk category within the current Mexican environmental legislation (SEMARNAT, 2019). This discrepancy poses a challenge for implementing local and regional conservation strategies, as national policies do not adequately reflect the urgency of protecting the species.
This study conducted a comprehensive review of the scientific literature available on D. mexicana, with the aim of compiling and analyzing information related to its distribution, ecology, and conservation status. A systematic search for scientific articles was carried out using databases such as Google Scholar, Scopus, Web of Science, SciELO, and AI-powered search engines such as ResearchRabbit and Semantic Scholar. The latter provided additional advantages by identifying non-obvious bibliographic connections through citation network analysis and suggesting relevant literature using machine learning algorithms, thereby helping to detect studies that might have been missed through traditional keyword-based searches (Rodas-Trejo, 2024).
The keywords used for the search included: “Dasyprocta mexicana”, “Mexican agouti”, “agutí mexicano”, and “sereque mexicano”. Additionally, the reference lists of the retrieved articles were reviewed to identify further relevant studies. Inclusion criteria comprised any studies that provided information on the species. Studies focusing solely on other Dasyprocta species were excluded. The information gathered was organized by Mexican state and synthesized in a comparative table displaying documented records of the species, type of record, abundance, and locality.
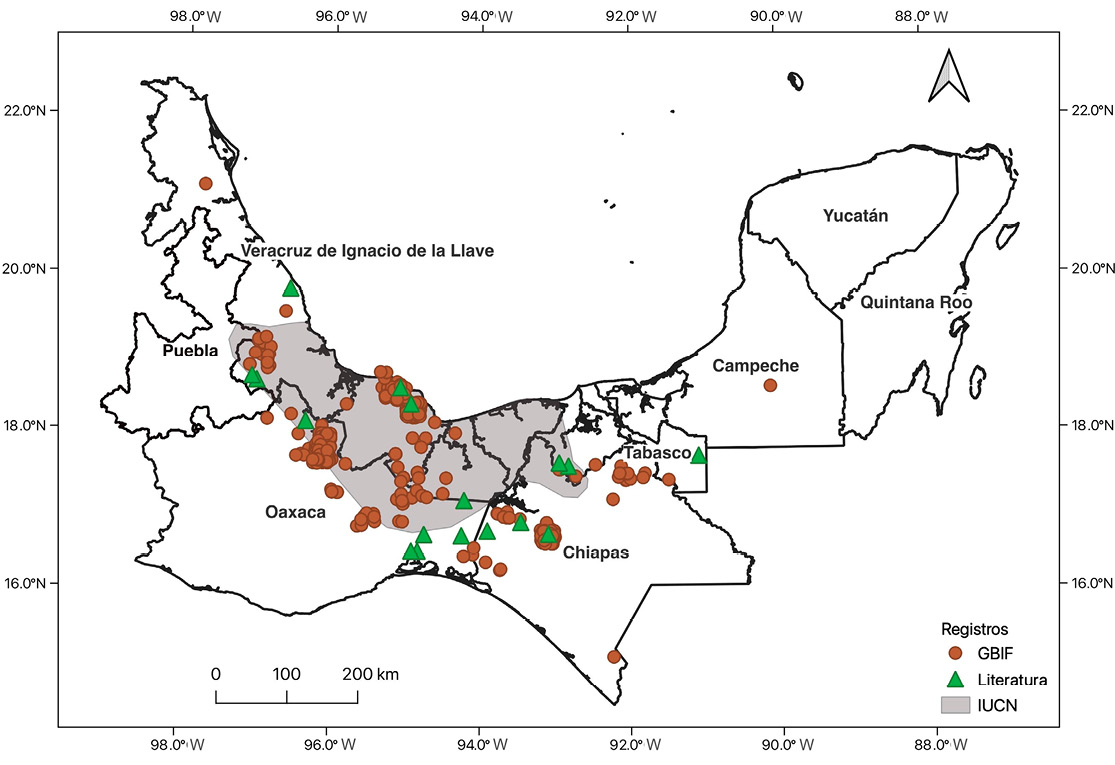
Furthermore, occurrence data for D. mexicana were retrieved from the Global Biodiversity Information Facility (GBIF), and the species’ distribution range established by the IUCN was reviewed (Roach et al. 2024). Based on this information, a distribution map was created, integrating documented GBIF records, IUCN range maps, and localities reported in the reviewed scientific literature.
The systematic review included 42 scientific articles reporting records of D. mexicana, complemented by 1,050 occurrence records obtained from the Global Biodiversity Information Facility (GBIF, 2025). The distribution of records by state revealed the highest concentration in Oaxaca (705 GBIF records; six localities in the literature), followed by Veracruz (246 GBIF records; five localities), Chiapas (84 GBIF records; three localities), Tabasco (14 GBIF records; three localities), and Campeche (one GBIF record; one locality). GBIF records were clustered in four main areas: the Sierra de Juárez in northern Oaxaca (110 records), the Sierra de Santa Martha and the Los Tuxtlas Biosphere Reserve in Veracruz (89 and 60 records, respectively), and the El Zapotal Ecological and Recreational Center in the city of Tuxtla Gutiérrez, Chiapas (57 records) (Figure 2).
Both GBIF data and scientific literature revealed records outside the species’ distribution range as defined by the IUCN, specifically in northern Chiapas near the municipality of Palenque, within the Selva El Ocote Biosphere Reserve, and in the southern region of Oaxaca. An additional record from the state of Campeche was documented, although it is likely the result of a misidentification.
Among the 42 reviewed studies, 23 reported sightings of D. mexicana as part of broader mammal community monitoring efforts. The remaining articles addressed topics such as the traditional use of mammals by rural communities, the status of endemic species in Mexico, interspecific interactions, and activity patterns of wildlife species. Only three studies focused specifically on D. mexicana.
Studies indicate that D. mexicana exhibits predominan-tly diurnal activity patterns, with records between 05:00 and 19:00 h, although crepuscular and nocturnal activity has also been observed (McWilliams 2009; Lira-Torres & Briones-Salas 2012; Ríos-Solís et al. 2021). A comparative study conducted in two protected areas in Chiapas demonstrated that although the species maintains a generally diurnal pattern, it may exhibit temporal variation in activity in response to differing levels of anthropogenic disturbance (Rodas-Trejo et al. 2025b). Regarding home range, Estrada-Croker and Naranjo-Piñera (1998) documented that D. mexicana occupies an estimated 3.65 hectares in El Zapotal Ecological and Recreational Center in Chiapas. From an ecological perspective, the species has been reported to act as both seed disperser and seedling predator, particularly for species such as Poulsenia armata (Asquith et al. 1999; Zambrano et al. 2015). It also serves as prey for mesopredators such as the ocelot (Leopardus pardalis) (Hernández-Sánchez et al. 2017).
Dasyprocta mexicana faces several threats, including deforestation, habitat fragmentation, and subsistence hunting (Sánchez-Cordero et al. 2005). The degradation of its natural habitat has resulted in a substantial loss of forest cover; it is estimated that the species has lost over one-third of its original habitat due to deforestation and land-use change, primarily from the conversion of forests to pastures, plantations, and agricultural fields (Sánchez-Cordero et al. 2005; Pedraza 2008). This habitat loss has increased the species’ vulnerability, particularly in the state of Oaxaca, where it is among the four most vulnerable endemic mammal species (Ramírez-Bautista et al. 2020). Although D. mexicana has traditionally been associated with well-preserved tropical forests, recent records in oil palm plantations and disturbed areas suggest a certain degree of ecological plasticity, which warrants further investigation (Franquesa-Soler et al. 2023).
In addition to habitat loss, subsistence hunting represents a significant pressure, as D. mexicana is one of the most frequently consumed wild mammals in the dry forests of the Mexican Pacific, in the Selva Zoque of Oaxaca, and in the Lacandon Jungle in Chiapas (Naranjo & Cuarón 2010; Lira-Torres & Briones-Salas 2011; Lira-Torres et al. 2012; Palacios-Silva et al. 2017). Furthermore, Lira-Torres and Briones-Salas (2012) documented that D. mexicana is also hunted due to the damage it causes to crops in the Selva Zoque region of Oaxaca.
In terms of health, Cucchi-Stefanoni et al. (2008) reported mortality cases caused by severe anemia due to infestation with fleas of the genus Echidnophaga. They also documented skin lesions characterized by dorsal alopecia and hyperplastic perivascular dermatitis, highlighting the species’ susceptibility to dermatological and hematological conditions associated with ectoparasites.
Research on D. mexicana remains limited. Most studies have focused on the states of Veracruz, Oaxaca, and Chia-pas, with few records from Tabasco (Santos-Moreno & Pérez-Irineo 2013; Pozo-Montuy et al. 2019; Zagal-García et al. 2022; Franquesa-Soler et al. 2023). Furthermore, there are very few studies exclusively dedicated to this species; most records derive from broader mammal community surveys, primarily using camera traps and sign surveys. Of the three studies specifically focused on D. mexicana, one estimated population size and density, feeding habits, effects on understory vegetation, and home range at the “El Zapotal” Ecological and Recreational Center (Estrada-Croker and Naranjo-Piñera 1998); another examined activity patterns by comparing an urban protected area and a wild area (Rodas-Trejo et al. 2025b), both conducted in Chiapas; and the third reported mortality associated with flea infestation (Cucchi-Stefanoni et al. 2008).
Regional Studies. In the state of Oaxaca, the presence of D. mexicana has been documented across various regions and ecosystems. In the Sierra Norte-Mixe, Hernández-Sánchez et al. (2017) recorded the species primarily in semi-evergreen forest, where it was positively associated with L. pardalis, suggesting its key role in the food web as a prey species. In contrast, Cortés-Marcial and Briones-Salas (2014) recorded a single individual in the Isthmus of Tehuantepec, marking the first documentation of the species in deciduous lowland forest. Meanwhile, in Santa María Chimalapa, within tropical evergreen forest, Santos-Moreno and Pérez-Irineo (2013) recorded five individuals.
In the Zoque Forest of Oaxaca, several studies have focused on D. mexicana. Lira-Torres and Briones-Salas (2012), along with Lira-Torres et al. (2012), made significant contributions to the understanding of the species in this region, where it was identified as the third most abundant species in the Chimalapas region. These authors demonstrated that D. mexicana prefers well-preserved forests, even in areas with hunting pressure, while avoiding secondary vegetation. The study also identified D. mexicana as an important species for local subsistence, as it is used as a source of animal protein (bushmeat) in the region.
Regarding its altitudinal distribution, Ramírez-Bautista et al. (2020) reported that in Oaxaca, the species occurs between 50 and 650 meters above sea level, indicating a relatively narrow elevational range of 600 meters, which may increase its sensitivity to climate change. Furthermore, this study identified D. mexicana as one of four species classified as “highly vulnerable” under the most severe climate change scenario projected for the late century (2075–2099). In Cerro Tepezcuintle, in the municipality of San Miguel Soyaltepec, Pérez-Irineo and Santos-Moreno (2012) found D. mexicana to be the third most frequently recorded species. Finally, in San Lorenzo Pápalo, located in the municipality of Concepción Pápalo, within the Cuicatlán Valley, Solís and Casas (2019) documented D. mexicana as one of the hunted species in a study assessing traditional ecological knowledge and wildlife use.
In the state of Veracruz, research on D. mexicana has been especially abundant in the Los Tuxtlas region. Flores et al. (2014), working in the Los Tuxtlas Biosphere Reserve, obtained 2,863 independent records, while Flores-Martínez et al. (2022) recorded 318 individuals, and also found a negative relationship between D. mexicana and mesopredators such as the ocelot (L. pardalis) and the coyote (Canis latrans). Similarly, Zagal-García et al. (2022) documented its presence during the rainy season in experimental ecological restoration plots, specifically in plantations composed of animal-dispersed plant species. Ríos-Solís et al. (2021) conducted a study on medium-and large-sized mammal diversity and activity patterns in the same reserve and reported D. mexicana as one of the species with the highest capture rate. They recorded a primarily diurnal activity pattern (67.1% of records), with some cre-puscular (25.6%) and nocturnal (7.3%) activity. Dasyprocta mexicana was described as a relatively common species in the tropical forests of the region. González-Christen and Coates (2019) also reported the species as part of the regio-nal mammalian fauna of the Los Tuxtlas Biosphere Reserve.
In other areas of Veracruz, Salazar-Ortiz et al. (2020) detected only three individuals in the municipality of Tequila, classifying the species among “rare mammals” and noting that these records represented new localities within its potential distribution range. In contrast, Gallina and González-Romero (2018), during monitoring efforts in the La Otra Opción Ecological Reserve, identified D. mexicana as the fourth most common species. In the Sierra de Zongolica, Mezhua-Velázquez and Bolívar-Cimé (2023) documented its presence and classified it among the species potentially affected by anthropogenic pressures, particularly from feral dogs. In the same area, Mezhua-Velázquez et al. (2022) reported D. mexicana as the most frequently recorded species, and identified three types of uses by local residents: for food, commercial trade, and sport hunting.
Peterson et al. (2000), in a study aimed at identifying conservation priorities for endemic species in Veracruz, listed D. mexicana among the eight priority endemic mammal species, with a distribution area estimated at 1,712 km². González Kuk and Neger (2020) documented the integration of D. mexicana into conservation initiatives with sustainable use, such as Wildlife Management Units (UMAs), specifically in “La Otra Opción” in Los Tuxtlas, where the species is promoted through ecotourism.
In the state of Chiapas, Estrada-Croker and Naranjo-Piñera (1998), in a study conducted at the El Zapotal Ecological and Recreational Center—where D. mexicana was accidentally introduced after escaping from an enclosure at the Miguel Álvarez del Toro Zoo (ZOOMAT)—reported population densities ranging from 3.2 to 5.4 individuals/ha. This research also provided dietary information, documenting the consumption of 44 plant species and ten fungal genera, with the latter representing the first recorded instances of mycophagy in this species. Lorenzo et al. (2017), in an assessment of the current status of terrestrial mammal diversity, distribution, and conservation in Chiapas, recorded D. mexicana in the Highlands and Sierra Madre of Chiapas, across various vegetation types, including tall, medium, and lowland tropical forests—evergreen, semi-evergreen, deciduous—as well as secondary vegetation and human settlements.
In the Selva El Ocote Biosphere Reserve, Pozo-Montuy et al. (2019) and Rodas-Trejo et al. (2025a) found D. mexicana to be the second most frequently recorded species after C. paca. However, these studies reported contrasting habitat preferences. While Pozo-Montuy et al. (2019) documented the species primarily in disturbed areas—suggesting a degree of adaptation to modified habitats—Rodas-Trejo et al. (2025a) found it to prefer less disturbed zones, located at lower elevations and further from major roads. Additionally, Riechers-Pérez (2004) reported its presence in the Laguna Bélgica Ecological Conservation Zone in Ocozocoautla.
In the state of Tabasco, Franquesa-Soler et al. (2023) documented the presence of D. mexicana in oil palm plantations within a highly deforested region near the municipality of Tenosique. The species was photo-recorded in two out of eleven plantations surveyed and also reported by farmers in two additional localities, suggesting a cer-tain degree of adaptability to anthropogenically modified environments. Gordillo-Chávez et al. (2023), on the other hand, recorded D. mexicana in the Sierra de Tabasco State Park, where it was documented across the three studied sierras (Madrigal, Poana, and Tapijulapa), both in medium-height evergreen forest and secondary vegetation.
Dasyprocta mexicana was introduced to Cuba in the 1930s alongside D. punctata and C. paca, in Sierra de Órganos and Sierra Cristal. It became established by 1967, primarily in the western part of the island—in San Diego, Soroa, and Guanahacabibes in Pinar del Río. Over time, its populations declined, with sporadic sightings reported in San Diego de los Baños between 1990 and 1993. In 2008, a specimen of D. mexicana was hunted near Soroa (Borroto-Páez 2009). However, contrasting these reports of population decline, Mancina et al. (2000) noted that the species was abundant in the Sierra del Rosario Biosphere Reserve.
Scientific literature reviewed documented D. mexicana in 17 different locations, mainly within protected natural areas, using primarily camera trap surveys (14 studies), complemented by direct observations and signs of presence. Detection rates varied widely—from a single record in the Isthmus of Tehuantepec to 2,863 records in the Los Tuxtlas Biosphere Reserve (Table 1). The information spans approximately three decades, from the work of Estrada-Croker and Naranjo-Piñera (1998) to the most recent contributions by Rodas-Trejo et al. (2025b).
Scientific knowledge on D. mexicana remains limited, with significant gaps in key areas such as detailed population studies, genetic analyses to confirm taxonomic identity, assessments of its adaptability to disturbed habitats, and determination of specific habitat requirements and connectivity needs. These knowledge gaps hinder the development of effective conservation strategies for a species that has lost over one-third of its natural habitat due to deforestation and land-use change, emphasizing the urgent need to increase research efforts, especially considering its critical conservation status and ecological role in southeastern Mexico’s tropical forests.
The discrepancy between the species’ classification as Critically Endangered by the IUCN and its absence from Mexican environmental legislation reflects methodolo-gical differences in risk assessment: while international evaluations rely on distribution models and global population trends, Mexican regulations use more restrictive criteria that often underestimate extinction risk for endemic species (García-Aguilar et al. 2017). This situation, combined with the urgent need to confirm its taxonomic validity through molecular analyses (Ruiz-García et al. 2022), highlights the importance of revising and updating national conservation policies to include D. mexicana in the NOM-059-SEMARNAT as a species at risk.
Advancing research focused on population demogra-phy, molecular genetics, habitat ecology, and responses to anthropogenic disturbances—along with the implementation of long-term monitoring programs—will be essential to generate the evidence-based knowledge needed to support conservation actions and resolve the taxonomic and ecological uncertainties currently limiting the species’ effective protection.
Acknowledgements
We thank the reviewers and editors for enriching our work with their feedback.
Literature cited
Asquith, N. M., et al. 1999. The fruits the agouti ate: Hymenaea courbaril seed fate when its disperser is absent. Journal Of Tropical Ecology 15;229-235.
Borroto-Páez, R. 2009. Invasive mammals in Cuba: an overview. Biological Invasions 11:2279-2290.
Cortés-Marcial, M., and M. Briones-Salas. 2014. Diversidad, abundancia relativa y patrones de actividad de mamíferos medianos y grandes en una selva seca del Istmo de Tehuantepec, Oaxaca, México. Revista de Biología Tropical 62:1433-1443.
Cucchi-Stefanoni, K., et al. 2008. Fatal anemia and dermatitis in captive agoutis (Dasyprocta mexicana) infested with Echidnophaga fleas. Veterinary Parasitology 155:336-339.
Estrada-Croker, J. C., and E. J. Naranjo-Piñera. 1998. Ecología del Agutí (Dasyprocta mexicana) en El Zapotal, Chiapas Instituto de Historia Natural del Estado de Chiapas, en Informe Final SNIB-CONABIO Proyecto No. G020. http://www.conabio.gob.mx/institucion/proyectos/resultados/InfG020.pdf
Franquesa-Soler, M., et al. 2023. Socioecological assessment of mammal assemblages in small oil-palm plantations in a highly deforested region in Mexico. Tropical Conservation Science 16:1-12.
Flores, J. J., et al. 2014. Mamíferos terrestres de la Estación de Biología Tropical de Los Tuxtlas. Revista Digital Universitaria 14:1-10. https://www.ru.tic.unam.mx/handle/123456789/2209
Flores-Martínez, J. J., et al. 2022. Spatiotemporal coexistence of mesopredators and their prey in a defaunated Neotropical rainforest. Tropical Conservation Science 15:1-11.
Gallina, S. and A. González-Romero. 2018. La conservación de mamíferos medianos en dos reservas ecológicas privadas de Veracruz, México. Revista Mexicana de Biodiversidad 89:1245:1254.
García-Aguilar, M. C., et al. 2017. La fauna nativa de México en riesgo y la NOM-059: ¿Están todos los que son y son todos los que están?. Acta Zoológica Mexicana (N S) 33:188-198.
GBIF.org (17 June 2025) GBIF Occurrence Download https://doi.org/10.15468/dl.e4nsuw
González-Christen, A., and R. Coates. 2019. Los mamíferos no voladores de la región de Los Tuxtlas, Veracruz, México. Revista Mexicana de Biodiversidad 90:1-15.
Gordillo-Chávez, E. J., et al. 2023. Mamíferos del Parque Estatal de la Sierra de Tabasco, Tabasco, México. Revista Bio Ciencias 10:e1461
Hernández-López, E., and H. M. J. López-Castilla. 2025. El pequeño dispersor de semillas de los ecosistemas. Therya Ixmana 4:95-96.
Hernández-Sánchez, A., A. Santos-Moreno and G. Pérez-Irineo. 2017. Abundance of mesocarnivores in two vegetation types in the southeastern region of Mexico. The Southwestern Naturalist 62:101-108.
International Union For Conservation Of Nature (IUCN). 2023. Dasyprocta mexicana. The IUCN Red List of Threatened Species. Version 2023-1.2. https://www.iucnredlist.org/. Accessed April 26, 2023.
Kuk, G. M. G., and C. Neger. 2020. El ecoturismo como estrategia de fortalecimiento en las acciones de conservación ambiental: Un análisis regional en Los Tuxtlas, Veracruz, México. PASOS Revista de Turismo y Patrimonio Cultural 18;571-584.
Lira-Torres, I. and M. Briones-Salas. 2011. Impacto de la ganadería extensiva y cacería de subsistencia sobre la abundancia relativa de mamíferos en la Selva Zoque, Oaxaca, México. Therya 2:217-244.
Lira-Torres, I. and M. Briones-Salas. 2012. Abundancia relativa y patrones de actividad de los mamíferos de los Chimalapas, Oaxaca, México. Acta zoológica mexicana (NS) 28: 566-585.
Lira-Torres, I., et al. 2012. Mamíferos de la Selva Zoque, México: riqueza, uso y conservación. Revista de Biología Tropical 60:781-797.
Lorenzo, C., et al. 2017. Diversidad y conservación de los mamíferos terrestres de Chiapas, México. Revista Mexicana de Biodiversidad 88:735-754.
Mancina, C. A., A. H. Marrero and L. R. Schettino. 2000. Mastofauna silvestre de la Reserva de la Biosfera «Sierra del Rosario», Cuba. Poeyana 9-13. https://biblat.unam.mx/es/revista/poeyana/articulo/mastofauna-silvestre-de-la-reserva-de-la-biosfera-sierra-del-rosario-cuba
Mcwilliams, D. A. 2009. Determinants for the diet of captive Agoutis (Dasyprocta spp.). Veterinary Clinics Of North America Exotic Animal Practice 12:279-286.
Mezhua-Velázquez, M. J., and B. Del S. Bolívar-Cimé. 2003. El temazate rojo: entre el desconocimiento y el desconocimiento y su aprovechamiento en las Altas Montañas de Veracruz, México. Therya Ixmana 2:21-22.
Mezhua-Velázquez, J. M., et al. 2022. Diversidad de mamíferos medianos y grandes del Ejido Zomajapa, Zongolica, Veracruz, México: implicaciones de manejo. Ecosistemas y Recursos Agropecuarios 9:1-15.
Naranjo, E. J. and A. D. Cuarón. 2010. Usos de la fauna silvestre. Pp. 271-283 in Diversidad, amenazas y áreas prioritarias para la conservación de las Selvas Secas del Pacífico de México (Ceballos, G., L. Martínez, A. Garcia, E. Espinoza, J. Bezaury and R. Dirzo, eds.). Fondo de Cultura Económica. D.F., México.
Palacios-Silva, R., et al. 2017. Inventario campesino de mamíferos medianos y grandes para un paisaje de la Selva Lacandona. Revista Mexicana de Mastozoología (Nueva Epoca) 1:25-34.
Pedraza, R. 2008. Compensar para conservar. Biodiversitas 76:1-16.
Pérez-Irineo, G. and A. S. Santos-Moreno. 2012. Diversidad de mamíferos terrestres de talla grande y media de una selva subcaducifolia del noreste de Oaxaca, México. Revista Mexicana de Biodiversidad 83:164-169.
Peterson, A., et al. 2000. Geographic analysis of conservation priority: endemic birds and mammals in Veracruz, Mexico. Biological Conservation 93:85-94.
Pozo-Montuy, G., et al. 2019. Análisis espacial y temporal de la estructura de la comunidad de mamíferos medianos y grandes de la Reserva de la Biosfera Selva El Ocote, en el sureste mexicano. Revista Mexicana de Biodiversidad 90:1-14.
Ramírez‐Bautista, A., et al. 2020. Trait‐based climate vulnerability of native rodents in southwestern Mexico. Ecology and Evolution 10:5864-5876.
Retana, O. G., and C. Lorenzo. 2002. Lista de los mamíferos terrestres de Chiapas: endemismo y estado de conservación. Acta zoológica mexicana (N S ), 85:25-49.
Riechers Pérez, A. 2004. Análisis mastofaunístico de la zona sujeta a conservación ecológica Laguna Bélgica, Chiapas, México. Anales del Instituto de Biología Serie Zoología 75:363-382.
Ríos-Solís, J. A., et al. 2021. Diversity and activity patterns of medium- and large-sized terrestrial mammals at the Los Tuxtlas Biosphere Reserve, México. Therya 12:237-248.
Roach, N., et al 2024. Dasyprocta mexicana. The IUCN Red List of Threatened Species 2024: e.T6285A22198164. Accessed on 23 June 2025.
Rodas-Trejo, J. 2024. Optimizando la búsqueda y análisis de artículos científicos en mastozoologíamediante el uso de plataformas académicas con inteligencia artificial. Revista Mexicana de Mastozoología Nueva Época 14:1-10.
Rodas-Trejo, J., et al. 2025a. Assessing the effects of landscape attributes in medium and large terrestrial mammal richness inside a tropical rainforest biosphere reserve. Environmental Monitoring and Assessment 197:1-19.
Rodas-Trejo, J. et al. 2025b. Patrones de actividad de Dasyprocta mexicana: contrastes entre un área urbana y un área silvestre en Chiapas, México. Mammalogy Notes 11: 492
Ruiz-García, et al. 2022. Mitogenomic phylogenetics and population genetics of several taxa of agouties (Dasyprocta sp., Dasyproctidae, Rodentia): molecular nonexistence of some claimed endemic taxa. Mammal Research 67:367–397
Salazar-Ortiz, J., et al. 2020. Diversidad de mamíferos del municipio de Tequila, Veracruz, México. Abanico Veterinario 10:1-18.
Sánchez-Cordero, V., et al. 2005. Deforestation and extant distributions of Mexican endemic mammals. Biological Conservation 4:465-473.
Santos-Moreno, A. 2014. Los mamíferos del estado de Oaxaca. Revista Mexicana de Mastozoología Nueva Época 4:18-32.
Santos-Moreno, A., y G. Pérez-Irineo. 2013. Abundancia de tepezcuintle (Cuniculus paca) y relación de su presencia con la de competidores y depredadores en una selva tropical. Therya 4:89-98.
Solís, L., and A. Casas. 2019. Cuicatec ethnozoology: traditional knowledge, use, and management of fauna by people of San Lorenzo Pápalo, Oaxaca, Mexico. Journal of Ethnobiology and Ethnomedicine 15:1-16.
Secretaría De Medio Ambiente y Recursos Naturales (Semarnat). 2010. Norma Oficial Mexicana NOM-059-SEMARNAT-2010. Protección Ambiental-Especies nativas de México de flora y fauna silvestres-Categorías de riesgo y especificaciones para su inclusión, exclusión o cambio-Lista de especies en riesgo. Diario Oficial de la Federación 2454:1-77. México.
Zagal-García, K. V., C. Martínez-Garza, and D. Valenzuela-Galván. 2022. Captura fotográfica de mamíferos medianos en parcelas experimentales de restauración ecológica en un paisaje agropecuario en Los Tuxtlas, Veracruz, México. Acta Botánica Mexicana 129:1-14.
Zambrano, J., R. Coates and H. F. Howe. 2014. Effects of forest fragmentation on the recruitment success of the tropical tree Poulsenia armata at Los Tuxtlas, Veracruz, Mexico. Journal of Tropical Ecology 30:209-218.
Zambrano, J., R. Coates, and H. F. Howe. 2015. Seed predation in a human-modified tropical landscape. Journal of Tropical Ecology 31:379-383.
Associate editor: Xavier López Medellín
Submitted: May 11, 2025; Reviewed: July 29, 2025
Accepted: August 07, 2025; Published on line: April 28, 2026